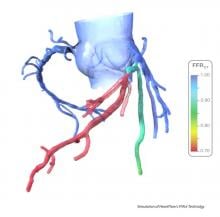
Fractional flow reserve-computed tomography (FFR-CT) is still in the early stages of clinical implementation in the United States, but it is already changing clinical practice as a non-invasive alternative to diagnosing patients with chest pain. Approved by the U.S. Food and Drug Administration (FDA) in November 2014, the technology provides both anatomical and functional assessment of the coronary arteries, a task no other method has accomplished to date.
DAIC spoke with three users of the HeartFlow FFR-CT technology, each in various stages of clinical practice: