
May 12, 2009 - The American College of Radiology (ACR) released a statement today saying that "Congress should act to require coverage of virtual colonoscopies."
The Congress is meeting today to review the recent determination by the Centers for Medicare and Medicaid Services (CMS) to not cover screening computed tomography colonography (CTC) exams, also known as virtual colonoscopy. This decision is not supported by the latest clinical evidence and may result in tens of thousands of unnecessary deaths each year from colorectal cancer, particularly among minority and underserved populations. CMS should reverse this determination and provide full coverage of CT colonography or Congress should pass legislation in this session to require Medicare coverage of the exam.
Recently, the landmark multi-center American College of Radiology Imaging Network Trial, sponsored by the National Cancer Institute and published in the New England Journal of Medicine, demonstrated that CT colonography is comparable in effectiveness to standard colonoscopy as a screening tool for the detection of cancer and precancerous polyps. The American Cancer Society has added CT colonography to its list of recommended colorectal cancer screening exams. The National Cancer Institute Colorectal Cancer Progress Review Group predicts that wider use of screening could save 20,000 lives annually.
“Make no mistake: If let stand, this CMS decision not to pay for CT colonography will cost lives. More than 140,000 Americans are diagnosed with colorectal cancer each year. Nearly 50,000 of them die due to late detection. How can CMS ignore the fact that people are dying because they do not want to have the tests that are currently covered?” said James H. Thrall, M.D., FACR, chair of the American College of Radiology Board of Chancellors. “For CMS to turn its back to a technology that can attract more patents to be screened and save countless lives is deeply concerning. CMS should reverse this determination immediately or Congress should step in and vote to mandate coverage of CTC.”
Colorectal cancer is the third most common cancer diagnosed among men and women in the United States and the second leading cause of cancer death, despite having a 90 percent cure rate when detected early. This is because less than half of those 50 and older who should be screened for the disease opt to receive the screening exams currently covered by Medicare.
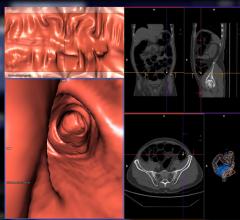
Noninvasive CT colonography uses cutting-edge x-ray technology to create three dimensional, fly-through images of the entire colorectal structure. The exam does not require sedation and patients may resume their normal daily activity immediately following. This has the ability to attract far more people to be screened and ultimately save lives.
“The percentage of patients at risk for the disease who have opted to be screened has not dramatically increased in more than 30 years. CTC is the technology that can overcome many stigmas associated with colorectal cancer screening, get more patients to be screened, and dramatically reduce the number of people who die unnecessarily each year from this disease,” said Elizabeth McFarland, M.D., chair of the ACR Colon Cancer Committee.
Major insurers CIGNA and UnitedHealthcare already cover screening CT colonography nationwide while Anthem Blue Cross Blue Shield provides coverage in certain states. Particularly in light of recent clinical trials affirming CT colonography’s accuracy, the CMS refusal to cover this exam in effect represents a reinforcement of a two-tier approach to coverage of screening for this deadly disease: one for those who have private insurance and lesser coverage for Medicare beneficiaries.
The CMS determination to not cover CT colonography will be particularly felt in minority and underserved populations. The Journal of the American Geriatrics Society released a study demonstrating that the disparity between white and non-white Medicare enrollees receiving a colonoscopy screening increased between 1995 and 2003.
“Providing Medicare patients an effective and less invasive screening tool for colorectal cancer can help overcome the disparity in colorectal care that exists in minority communities. This noncoverage determination is a setback to efforts to provide better care to underserved areas of the population. Congress needs to act now to help minority and underserved populations gain access to this potentially life saving care,” said McFarland.
For more information: www.acr.org

 February 06, 2024
February 06, 2024 








