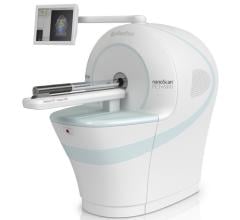
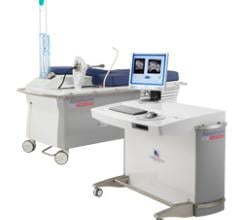
Dune Medical Devices announced U.S. Food and Drug Administration (FDA) approval of an updated version of its flagship product, MarginProbe, a medical device that enables real-time detection of cancer at the surface of excised tissue specimens during breast-conserving cancer surgery.
Carestream is now the first company approved to provide the latest picture archiving and communications systems (PACS) for use in U.S. Navy medical facilities as a result of meeting some of the most stringent product security requirements for the Navy’s DIACAP certification process.
Radiologists can transmit their reports, which can also contain access to images, to their referring physicians’ electronic health records (EHR) systems securely, efficiently, and cost effectively using carefully selected technology, according to a presentation by Advanced Data Systems Corp. (ADS) at the 2014 Radiology Society of North America (RSNA) annual meeting in Chicago, Nov. 30-Dec. 5.
Fujifilm’s APERTO Lucent is a 0.4T mid-field, open MRI system addressing today’s capability and image quality needs ...
Blackford Analysis announced that its software has been successfully integrated into a tool developed by Massachusetts General Hospital’s Tumor Imaging Metrics Core for its Precision Imaging Metrics system that helps radiologists and oncologists coordinate to generate clinical trial imaging results with enhanced efficiency and quality.
RaySearch Laboratories AB (publ.) has entered into a long-term distribution agreement with Kamol Sukosol Electric Co. (KEC), in Bangkok, Thailand.

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
Mediso Ltd. announced at the annual congress of the European Association of Nuclear Medicine the launch of new MRI subsystems with 3 and 7 Tesla field strength.
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
At the IEEE medical Imaging Conference (MIC) in November, ContextVision co-presented with Texas Instruments and High Performance Consulting on new research on 3-D adaptive filtering.
EDAP TMS SA received a letter from the Center for Devices and Radiological Health (CDRH) of the Food and Drug Administration (FDA) on its current premarket approval (PMA) application for the Ablatherm integrated imaging device for high-intensity focused ultrasound (HIFU).
A new surgical device enables breast cancer patients to be treated with dramatically improved targeting of radiation. Findings were presented at the 18th SIS World Congress on Breast Healthcare. The congress was co-hosted by the Senologic International Society (SIS) and the American Society of Breast Disease (ASBD).
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
Research and Markets has announced the addition of the "MediPoint: Nuclear Imaging - PET and SPECT Equipment - Global Analysis and Market Forecasts" report to their offering
Supplemental ultrasound screening for all U.S. women with dense breasts would substantially increase healthcare costs with little improvement in overall health, according to senior author Anna Tosteson, ScD, at Dartmouth Hitchcock's Norris Cotton Cancer Center and The Dartmouth Institute for Health Policy and Clinical Practice.
The value of volumetric breast density to help improve breast cancer risk prediction models and monitor clinical treatment was the focus of numerous abstracts presented at the 2014 San Antonio Breast Cancer Symposium (SABCS).
Did you know that approximately one-third of all the data in world is created by the healthcare industry and that ...
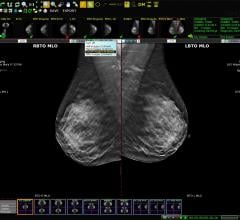
The value of volumetric breast imaging data to provide critical insight for optimal breast imaging quality and workflow was the focus of numerous abstracts accepted for presentation at the 100th Annual Meeting of the Radiology Society of North America (RSNA), November 30-December 5, 2014.
Viztek announced upgraded features that have been added to its Exa PACS.
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
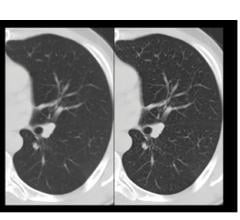
Less than 30 days after the Sapheneia Scannerside partnership announcement, the two companies released CMS Lung Screening, ClearCT Lung.
IDS debuted at RSNA 2014 AbbaDox CRM version 2.0 with advanced architecture, enhanced database design, intuitive graphical interface, geographic mapping functionality, analytics and a marketing toolset.
Blackford Analysis announced a technology partnership with Viztek.
Two-thirds of women treated for early-stage breast cancer in the United States receive longer radiation therapy than necessary, according to a new study published in the Journal of the American Medical Association (JAMA) from Penn Medicine researchers Ezekiel J. Emanuel, M.D., Ph.D., and Justin E. Bekelman, M.D.
Bayer HealthCare announced that the U.S. Food and Drug Administration (FDA) has approved Gadavist (gadobutrol) injection for use with magnetic resonance imaging (MRI) in pediatric patients less than 2 years of age, including term neonates, to detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system.
TiaLinx Inc. announced the launch of its new product line, the AR60-A.

 January 09, 2015
January 09, 2015