Fujifilm’s APERTO Lucent is a 0.4T mid-field, open MRI system addressing today’s capability and image quality needs ...
The need for radiology departments and imaging facilities to tap advancing technology while containing costs will continue to be a challenge. To succeed, the smartest strategy is to invest in imaging solutions that will adapt to your needs as you progress along the imaging continuum. Read the white paper to learn more.

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
Carestream now offers an oncology reading workflow for positron emission tomography (PET)/computed tomography (CT) studies.
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
May 5, 2016 — The National Basketball Association (NBA) and GE Healthcare announced the first round of winning research ...
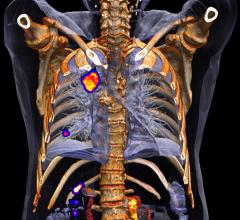
Doctors and researchers at Barnes-Jewish Hospital and Washington University in St. Louis may be able to better diagnose and monitor diseases at a functional level with GE Healthcare’s next-generation SPECT/CT system, Discovery NM/CT 670 CZT.
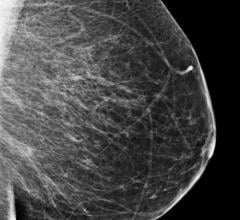
Only one in three women participating in Germany's mammography screening program (MSP) is well-informed about it: the higher the level of education, the greater the chance of women making an informed decision.
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
Eizo Inc. recently announced that it received U.S. Food and Drug Administration (FDA) 510(k) clearance for breast tomosynthesis for its 8 megapixel multimodality monitor, the RadiForce RX850.
Did you know that approximately one-third of all the data in world is created by the healthcare industry and that ...
May 4, 2016 — Infertility and hormonal fertility treatments may influence the amount of dense tissue in the breast, a ...
Today Siemens Healthcare unveiled its new brand name, Siemens Healthineers. The company said the new brand underlines Siemens Healthcare’s pioneering spirit and its engineering expertise in the healthcare industry.
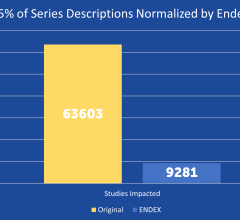
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
May 2, 2016 — In a new report, the U.S. Food and Drug Administration (FDA) suggests that since the passage of the ...
Infinitt North America announced that the company was highlighted in the new KLAS specialty report, “Imaging IT Landscape 2016 Enterprise Platform: The Next Nirvana?”
The U.S. Food and Drug Administration (FDA) announced it is building the foundations of a national evaluation system to generate better evidence more efficiently for medical device evaluation and regulatory decision-making.
Medicine supposedly aches for a universal viewer, one that can satisfy the needs of every physician — cardiologist, radiologist, dermatologist, orthopedist; and display its images on whatever device — laptop, desktop, tablet or smartphone.
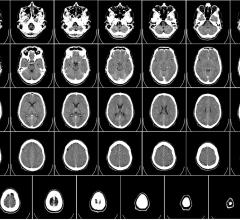
With increased use of computed tomography (CT) scans, radiologists and other experts — including Donald Frush, M.D., medical director of the Duke Medical Radiation Center and a co-author of a new World Health Organization (WHO) report — are urging awareness and accountability around medical scans for children.
The global market for contrast media injectors is set to increase from $830 million in 2015 to almost $1.8 billion by 2022, according to research and consulting firm GlobalData.