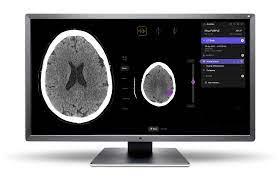
August 9, 2023 — Annalise.ai, one of the global leaders of AI decision-support solutions, announced that their Annalise Enterprise CTB for interpreting non-contrast head CT studies has received multiple additional regulatory approvals. Annalise Enterprise CTB is now CE marked as class IIb under EU Medical Device Regulation (EU MDR) and additionally has been approved for clinical use in Singapore.
The EU MDR and Singapore regulatory approvals signify a remarkable milestone for Annalise.ai, reaffirming the company's dedication to meeting the highest standards of safety and effectiveness. These certifications extend to the chest X-ray solution Annalise Enterprise CXR, highlighting the company's commitment to delivering cutting-edge AI technology to global healthcare markets through its comprehensive Enterprise AI platform.
"These regulatory approvals demonstrate our commitment to delivering top-quality, compliant, and innovative AI solutions to our valued customers and prospects. We are excited to enter the EU and Singapore markets. This is a significant step forward in our mission to improve patient outcomes globally through AI-driven healthcare," said Lakshmi Gudapakkam, CEO of Annalise.ai.
Annalise Enterprise CTB detects up to 130 radiological findings on non-contrast CT Brain (NCCTB) studies, including a wide range of conditions that require rapid intervention. It has been shown to improve diagnostic accuracy by 32%, averaged across all analyzed findings. Annalise Enterprise CXR can detect up to 124 findings on chest X-rays in under 20 seconds. It has been shown to improve accuracy by 45%, averaged across all analyzed findings2. The solutions allow worklist prioritization which supports reporting workflow efficiency by marking critical or unremarkable cases, according to clinical needs.
The latest round of regulatory approvals adds to a growing list for Annalise.ai, which has rapidly expanded its global footprint since its inception in 2020. Annalise Enterprise solutions are used in public and private healthcare in Australia, the United Kingdom, the United States, and throughout South East Asia. More approvals are anticipated as the company continues pursuing its mission to raise the standard of healthcare for millions of patients with cutting-edge, clinician-friendly AI technology.
For more information: www.annalise.ai

 August 29, 2024
August 29, 2024