Carl Bjartmar
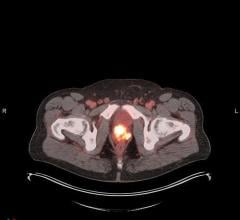
December 7, 2021 — Orviglance* is a novel manganese-based oral contrast agent for magnetic resonance imaging (MRI) developed to improve the detection and visualization of focal liver lesions (including liver metastases and primary tumors) in patients with reduced kidney function. These patients are at risk of serious, and potentially fatal, side effects from the currently available class of gadolinium-based contrast agents, which all carry black box warnings. Orviglance, which has been granted an Orphan Drug Designation by the U.S. Food and Drug Administration (FDA), is currently in Phase 3 development.
The comparison study versus the gadolinium contrast agent showed that Orviglance enhanced MRI was as effective as the gadolinium contrast agent gadobenate dimeglumine (Multihance) in terms of visualization of lesions and number of detected lesions. In fact, two out of the three independent readers reported higher scores for Orviglance-enhanced MRI. The results also showed that Orviglance-enhanced MRI provides improved diagnostic efficacy compared to MRI without a contrast agent using identical endpoints, as in the ongoing pivotal Phase 3 study, SPARKLE.
SPARKLE is a global multicenter study of Orviglance in up to 200 patients with severe rena impairment and known or suspected focal liver lesions. Primary efficacy, in terms of lesion visualization compared to unenhanced MRI (MRI without a contrast agent), will be evaluated by three independent blinded readers. Two supporting studies aim to provide further data about the use of Orviglance in clinical practice: one study includes patients with hepatic impairment (impaired liver function) and one study to determine the effect of food intake on Orviglance uptake, for which patient recruitment has been completed.
Carl Bjartmar, chief medical officer at Ascelia Pharma, said: “We are very pleased to be presenting the study as an oral paper presentation at the prestigious Radiological Society of North America’s radiology conference, RSNA. The study provides robust evidence of the diagnostic value that Orviglance can offer once it is available to patients and physicians. Orviglance is being developed to address the unmet medical need of patients with poor kidney function who require liver imaging, and it is very encouraging that Orviglance is as effective as a gadolinium contrast agent.”
The study was presented at the RSNA on Dec. 1, 2021, by Dr. Kohkan Shamsi.
For more information: www.ascelia.com
*Trademark is registered in Europe and several other markets and submitted for registration in the U.S.
Related Content of MRI Gadolinium Safety Concerns
The Debate Over Gadolinium MRI Contrast Toxicity
VIDEO: How Serious is MRI Gadolinium Retention in the Brain and Body? An interview with Max Wintermark, M.D.
VIDEO “Big Concerns Remain for MRI Gadolinium Contrast Safety at RSNA 2017,” An interview with Emanuel Kanal, M.D.
Radiology Has Failed to Properly Assess or Track MRI Gadolinium Contrast Safety
Recent Developments in Contrast Media
FDA Committee Votes to Expand Warning Labels on Gadolinium-Based Contrast Agents
European Medicines Agency Issues Update on Gadolinium Contrast Agents
ISMRM Issues Guidelines for MRI Gadolinium Contrast Agents
FDA: No Harm in MRI Gadolinium Retention in the Brain
VIDEO: MRI Gadolinium Contrast Retention in the Brain
Gadolinium May Remain in Brain After Contrast MRI

 July 30, 2024
July 30, 2024