October 27, 2008 - TeraRecon Inc. is highlighting to its CT Colon package at the 9th International Symposium on Virtual Colonoscopy held October 27-29, 2008 in Boston, MA.
The New England Journal of Medicine, in September 2008, published results from the recent National CT Colonography (CTC) multicenter trial. The study, conducted by the American College of Radiology Imaging Network (ACRIN) at 15 centers across the U.S, showed very comparable results as compared to the gold standard screening method of Optical Colonoscopy (OC). The study compared primary CTC screening with OC screening in 2,531 adults and found that primary CTC when used to screen asymptomatic patients has 90 percent sensitivity for polyps 10 mm or larger.
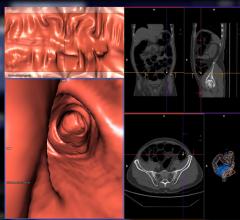
TeraRecon's 3D solution delivers a CT Colon package that features tools for confidence in diagnosis. The application provides automated segmentation and centerline generation for endoscopic evaluation. Also included are supine and prone study synchronization, rear-view mirror view, 100% visualization verification and single click polyp identification. An optional Spherefinder module automatically highlights regions of potential interest. The clinical application also supports iCAD or MedicSight Computer Assisted Detection (CAD) software that automatically identifies colon polyps and provides the clinical end-user with the ability to perform “concurrent” or “secondary reads” that can aid in the interpretation process. A powerful integrated reporting feature completes the interpretation process, producing reports which can be stored as a DICOM object and distributed to PACS.
Aquarius iNtuition addresses image management and workflow needs over enterprise networks. In this era of tightly-controlled healthcare costs, TeraRecon believes the client-server solution offers the optimal value without sacrificing performance or functionality. iNtuition brings necessary tools to everyone involved in the patient care cycle to achieve an accurate diagnosis and treatment plan quickly and efficiently. Each stakeholder along the imaging pathway can have access to the data in their own environment and customize their own workflow to achieve success. Architecturally speaking, the iNtuition platform offers flexibility, scalability and affordability, allowing imaging practitioners to design and deploy a solution uniquely suited to their specific needs in terms of rendering capacity and number of users.
Another unique feature of Aquarius iNtuition, is AquariusWEB, a browser-based viewer that delivers live, interactive, 3D reports to the interventionalists and surgeons who are the ultimate end-users of the imaging data. AquariusWEB gives the possibility to the physicians to simply open a URL link sent via an email or embedded in the patient record and have instant access to review and interact with the volumetric dataset. No plug-in or installation is required. AquariusWEB works on a standard web browser such as Internet Explorer, Firefox, or Chrome, as well as Mac and Unix-based browsers. AquariusWEB also features a mobile edition compatible with the iPhone.
Aquarius iNtuition is designed to bridge the clinical workflow and dataflow gaps between image acquisition, post-processing, interpretation and diagnosis and is uniquely capable of delivering best-in-class advanced visualization capabilities throughout an entire healthcare enterprise.
For more information: www.terarecon.com

 February 06, 2024
February 06, 2024 








