November 17, 2016 — The American Society for Radiation Oncology (ASTRO) issued an updated clinical practice statement for accelerated partial breast irradiation (APBI) for early-stage breast cancer. The updated guideline reflects recent evidence that greater numbers of patients can benefit from accelerated treatment, and also provides direction for the use of intraoperative radiation therapy (IORT) for partial breast irradiation.
The update to the 2009 ASTRO consensus statement for APBI is available as a free access article in Practical Radiation Oncology, ASTRO’s clinical practice journal.
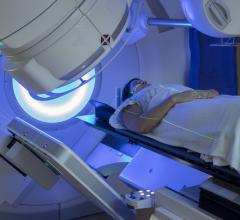
Early-stage breast cancer patients often receive radiation therapy (RT) following breast-conserving surgery to lower the chance of recurrence or metastasis, generally in the form of whole breast irradiation (WBI) using external beam radiation delivered over several weeks. APBI is a newer, localized alternative that delivers the effective radiation dose directly to the tissue at risk. This targeted therapy reduces treatment time and may limit adverse side effects, particularly those involving the heart and lungs. More than 75,000 women in the United States have received APBI since its introduction in the late 1990s.
Drawing on evidence published in the last eight years, the new guideline suggests that more breast cancer patients can benefit from APBI, including younger patients and those with low-risk ductal carcinoma in situ (DCIS). The guideline recommends considering the following factors for the use of APBI outside of clinical trial settings:
- Age: Patients age 50 or older should be considered suitable for APBI; patients age 40-49 who meet other pathologic suitability criteria should be considered cautionary; and patients younger than 40 or those who do not meet the pathologic criteria should be considered unsuitable. The age cohorts in the original guideline were = 60, 50-59 and < 50, respectively;
- DCIS status: Patients with low-risk DCIS should be considered suitable for APBI if they meet all aspects of the definition of “low-risk” DCIS from RTOG 9804, including screen-detected disease, low to intermediate nuclear grade, tumor size = 2.5 cm and surgical resection with margins negative at = 3 mm. The original guideline did not place any DCIS patients in the suitable group; and
- Surgical margins: There were no changes, maintaining the current standards that patients are suitable if surgical margins are negative by = 2 mm; cautionary if margins are close or < 2 mm; and not suitable if margins are positive.
The update also provides guidance for the use of intraoperative RT for early-stage breast cancer patients, drawing on two large, phase III clinical trials that compared WBI with IORT. Partial breast irradiation may be administered in a variety of methods, including brachytherapy, external beam RT and IORT, a single-dose radiation treatment commonly delivered at the time of surgery using either an electron beam or low-energy X-rays. Recommendations for the use of IORT include:
- Physicians should counsel patients that the risk of ipsilateral breast tumor recurrence (IBTR) may be higher with IORT compared with WBI, based on evidence from both existing clinical trials;
- Electron beam IORT should be restricted to patients with invasive cancer who are also considered suitable for partial breast irradiation, based on data from a trial with 5.8-year median follow-up;
- Low-energy X-ray IORT should be used only in the context of a prospective registry or clinical trial and restricted to women with invasive cancer who are considered otherwise suitable for partial breast irradiation. This recommendation reflects the short, 2.4-year median follow-up of existing trial data; and
- Given the persistent risk of IBTR over time, all patients treated with IORT should undergo routine long-term follow-up for at least 10 years to screen for tumor recurrence.
“As trials mature and evidence accumulates, we can understand more comprehensively who benefits from accelerated radiation treatment following lumpectomy, and we are finding that the pool of suitable candidates for this emerging treatment is larger than first anticipated,” said Jay R. Harris, M.D., FASTRO, chair of the task force that developed the guideline and distinguished professor of radiation oncology at the Dana-Farber Cancer Institute, Harvard Medical School in Boston. “Carefully selected patients may achieve similar tumor control following shorter, targeted schedules of radiation as they would with weeks of radiation to the whole breast.”
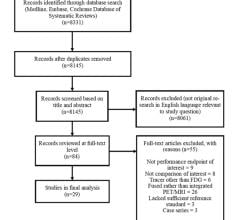
The guideline was based on a systematic literature review of studies published since May 2008, the last date searched for the original clinical practice statement. A total of 419 abstracts were retrieved from PubMed, and the 44 articles that met inclusion criteria were abstracted into evidence tables and evaluated by an eight-member task force of radiation oncologists, medical oncologists and experts in IORT. The clinical practice statement was approved by ASTRO’s Board of Directors following a four-week period of public comment.
For more information: www.practicalradonc.org

 August 09, 2024
August 09, 2024