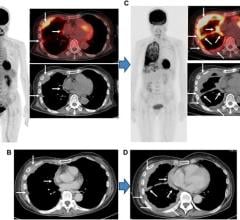
SNM's Clinical Trials Network (CTN) last week validated its 100th scanner, an important milestone in CTN's two-and-a-half year history. Scanner validation is an essential component of the CTN imaging site qualification program, which works with sites to ensure high-quality imaging capability by assessing both equipment and personnel capabilities.
"The validation of over 100 scanners in less than three years demonstrates the strength, viability and maturity of the CTN program," said Paul Christian, BS, CNMT, PET, chairman of the CTN scanner validation committee. "These validated scanners provide standardized image quality, lesion detectability and quantitative accuracy of the PET/CT scans performed on these systems. Drug companies can rely on the accuracy of images and standardized uptake value measurements from these scanners."
This unique international program ensures PET/CT scanner performance for sites involved in multicenter imaging trials. To have a scanner validated, imaging sites must first complete a one-page imaging equipment form and submit to CTN staff. The CTN scanner validation committee reviews the information to assess imaging equipment capabilities. If acceptable, the unique CTN anthropomorphic phantom and accompanying instructions are sent to the site. Images and completed forms are then returned to the CTN Core Lab for review. Once deemed acceptable, the site will receive a Scanner Validation Certificate for each validated scanner.
"Achieving scanner validation is a major step for imaging sites interested in participating in multicenter clinical trials,” said Christian. “Having the CTN seal-of-approval instills confidence in drug developers that a site will produce high-quality, reproducible images.”
In 2008, SNM formed CTN to facilitate the use of molecular imaging biomarkers in multicenter clinical trials. Since its launch, CTN has worked to provide drug developers with access to SNM's multicenter investigational new drug application for fluorothymidine, to qualify sites and validate scanners of imaging sites, and to provide nuclear medicine and molecular imaging professionals with specific imaging research education.
"It's not just drug developers and research-based imaging sites that benefit from participation in CTN," said Michael Graham, Ph.D., M.D., co-chair of CTN. "The more molecular imaging is standardized in clinical trials, the better the data. Better data will lead to greater acceptance, and, consequently, further development and clinical use of new biomarkers. The entire field benefits from the work of CTN."
For more information: www.snm.org/ctn

 July 30, 2024
July 30, 2024