January 6, 2012 – Bracco Diagnostics said it plans to close out the recall process for its CardioGen-82 cardiac perfusion radiotracer with a list of actions to bring the product back to market.
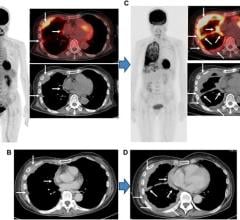
The company instituted the product recall on July 29, 2011 after two cardiac patients who underwent positron emission tomography (PET) scans set off radiation detectors on the U.S. border. The increased level of radiation was due to a strontium (Sr) breakthrough from the radiotracer generator.
In a letter to customers released December 29, Bracco said during the past five months it performed the following actions:
• CardioGen-82 Generator Voluntary Recall Process: All recalled generators were sent back to the manufacturer for functional and breakthrough testing. The generator testing demonstrated that each and every generator had Sr-82 and Sr-85 levels well within specification limits set forth in the prescribing information.
• Clinical Assessment Program: Bracco continues to gather data in a clinical study (designated CGEN-105) at all participating institutions that have administered CardioGen-82 to patients from January 2011 through July 2011. To date, there have been no patients identified at these sites with higher than expected radiation levels.
• Rb82 Generator Quality Review Program: Bracco initiated an on-site review of each facility’s daily quality control records, using the quality review process. This review was an opportunity to reinforce customer training and education and will be the basis for enhanced CardioGen-82 training quality control assessment and monitoring upon reintroduction of CardioGen-82 to the market.
• Manufacturing Process Review: At the U.S. Food and Drug Administration’s (FDA) request, Bracco initiated the re-qualifying of all CardioGen components and revalidating of the manufacturing process.
• Enhanced Labeling and User Training: Bracco continues to work with the FDA on enhancing the CardioGen-82 prescribing information and user training parameters to ensure proper field use. We will initiate a new quality control data repository and monitoring program upon the reintroduction of Cardiogen-82 to the market.
The company said it proposed to FDA a controlled and phased reintroduction of CardioGen-82 generators to user facilities, with data collection and evaluation of the actual field use of each generator beginning in the first quarter of 2012. Bracco said the FDA expressed support for this plan. The first step in this effort will be to implement customer requirements in advance of commercial generator availability. Bracco’s field nuclear medicine team will begin to contact each facility toward the end of January, discussing compliance training and site preparation expectations. Specific customer generator delivery schedules are not available at this time. However, customers can anticipate receiving additional information regarding generator availability by Feb. 1, 2012.
For more information: www.cardiogen.com

 July 30, 2024
July 30, 2024