June 29, 2017 — Philips announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market IntelliSpace Portal 9.0 and a range of innovative applications for radiology in the United States. The latest release of Philips’ clinical informatics platform for advanced visual analysis and quantification of medical images now offers enhanced additional applications for longitudinal brain imaging and multimodality tumor tracking, as well as optimized lung nodule assessment.
IntelliSpace Portal 9.0 has been available outside of the U.S. since November 2016 and will be available in the U.S. as of this month.
The new innovations that are now cleared add to more than 70 applications that IntelliSpace Portal 9.0 offers on a single platform, spanning clinical domains within radiology, including neurology, oncology and cardiology. The platform gives clinicians a comprehensive overview of each patient, helping them to quantify and quickly diagnose conditions using multimodality clinical applications that are optimized for patient evaluation over longer periods of time.
The number of people diagnosed with cancer and chronic diseases such as cardiovascular disease, chronic obstructive pulmonary disease (COPD) and dementia continues to rise. Timely diagnosis and early intervention can improve patient outcomes. These conditions also require longer-term treatment and continued follow-up to monitor a patient’s response to a chosen therapy over time and define the next steps in the treatment process.
“Analytics applications optimized for clinical decision support and longitudinal and quantified patient tracking are becoming increasingly important to radiologists,” said Mark van Buchem, professor of neuroradiology at the Leiden University Medical Center, one of the development partners of IntelliSpace Portal 9.0. “They can help visualize and quantify very subtle manifestations of disease and differences over time that may not be seen with the naked eye. IntelliSpace Portal 9.0 integrates into our existing workflow and adds greatly to our patient care.”
The newest clinical advance on the Intellispace Portal now cleared for U.S. distribution is its Longitudinal Brain Imaging (LoBI) – an application to analyze brain images to support the evaluation of neurological disorders over time. LoBI helps clinicians monitor disease progression in patients with neurodegenerative disorders such as stroke, Alzheimer’s disease and multiple sclerosis (MS).
The qEASL (quantitative European Association for the Study of the Liver) capability within Multi-Modality Tumor Tracking offers a new method for enhanced measurement of tumor volume based on magnetic resonance imaging (MRI) and computed tomography (CT) scans. This functionality aims to improve the current standard for cancer treatment follow-ups by giving a visual indication of how cells respond to therapy.
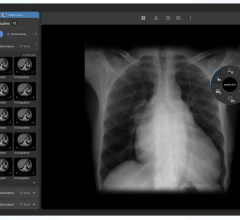
Also now available in the U.S. is Lung Nodule Assessment, which offers a diagnostic patient imaging tool that provides quantitative and characterization information about nodules in the lung in a single CT study, or over time during the course of multiple thoracic studies.
For more information: www.usa.philips.com/healthcare


 February 03, 2025
February 03, 2025