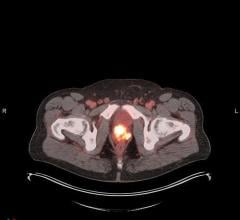
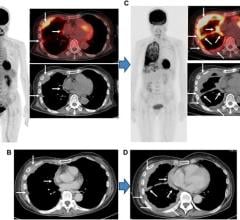
May 27, 2020 — PETNET Solutions, Inc., a Siemens Healthineers company, and Zionexa USA, a wholly owned subsidiary of Zionexa SAS, have announced that the Food and Drug Administration (FDA) has approved Cerianna (fluoroestradiol F 18) injection for intravenous use. Cerianna (fluoroestradiol F 18) is a molecular imaging agent indicated for use in positron emission tomography (PET) imaging for the detection of estrogen receptor-positive lesions as an adjunct to biopsy in patients with recurrent or metastatic breast cancer. Cerianna (fluoroestradiol F 18) is the first FDA-approved F-18 PET imaging agent specifically indicated for use in patients with recurrent or metastatic breast cancer.
Cerianna (fluoroestradiol F 18) will be commercially available beginning in late 2020/early 2021 through PETNET Solutions, Inc., Zionexa USA’s manufacturer and exclusive commercial distributor in the U.S. Additional manufacturing sites will be added as each site receives regulatory approval to commence manufacturing.
“PETNET Solutions, Inc., is proud to work with Zionexa USA as the exclusive U.S. commercial supplier making Cerianna (fluoroestradiol F 18) accessible to imaging centers and their breast cancer patients,” said Barry Scott, head of PETNET Solutions, Inc. “Our extensive network of radiopharmacies enables us to increase access to cutting-edge radiotracers such as Cerianna (fluoroestradiol F 18), helping healthcare facilities address the challenge of recurrent and metastatic breast cancer.”
“Zionexa is pleased to be able to make Cerianna (fluoroestradiol F 18) commercially available through the extensive manufacturing network of PETNET Solutions, Inc.,” said Peter Webner, chief executive officer of Zionexa USA. “PETNET has been a great partner to Zionexa and has surpassed our expectations as a contract manufacturer. Cerianna (fluoroestradiol F 18) will provide clinicians with additional, previously unavailable data on the estrogen receptor status of tumors across the patient’s entire body, providing additional data to enhance therapeutic decision-making.”
For more information: www.zionexa.com/


 July 31, 2024
July 31, 2024