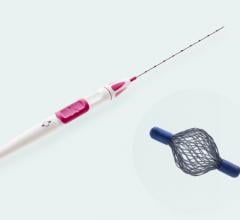
The Visica 2 Treatment System is an effective alternative to surgery for benign breast tumors
Until a year ago, I was treating biopsy-proven fibroadenomas by either observation or surgical excision. While I find surgical excision effective, the drawbacks include the need for general anesthesia, increased patient stress, recovery time, and based on fibroadenoma size — scarring and potential disfigurement. Observation can also be stressful on patients, especially when their fibroademomas are larger than 1 centimeter. Wanting a minimally invasive alternative for my patients with symptomatic fibroadenomas, I identified the Sanarus Visica 2 System that utilizes cryoablation to destroy fibroadenomas. After 12 months of use in my practice, I have found this therapy to be ideal in treating these symptomatic fibroadenomas from both the physician and the patient perspective.
The first advantage of cryoablation over surgery that I noted is that I can treat fibroadenomas quickly and easily in my office. This makes scheduling easier and has helped facilitate more treatments while reducing overall costs to my patients as co-pays are less. When performing Visica treatments, I use ultrasound to guide the cryoprobe into position in the lesion and to monitor a growing ice ball that engulfs the fibroadenoma as it moves through freeze/thaw cycles which destroy the targeted tissue. The Visica 2 treatment also affords me the opportunity to control the iceball size relative to the size of the lesion, giving me the ability to tailor my treatments to specific lesion sizes. The speed of the treatment was surprising. I’ve found treatment times for lesions 2 centimeters to be around 12 minutes. That combined with the use of local anesthesia makes the treatment stress-free on my patients.
Long-term results have also been impressive. Clinical studies have shown no regrowth in over 1,500 cases, which has also been demonstrated in my practice. Resolution has been rapid, especially with tumors under 2 centimeters in size that resolve in less than six months. Additionally, published clinical data demonstrates that cryotherapy does not produce artifacts in follow-up mammograms that would adversely affect interpretation. I have found follow-up mammography to be essentially normal in my patients.
Overall, I would describe the key difference between treating with cryotherapy versus surgical and percutaneous excision as the ability to perform a minimally invasive treatment that destroys the entire fibroadenoma with little or no risk. The ablated tissue essentially “melts away” over time, permanently ridding my patients of the fibroadenoma. They are left with virtually no scarring and no deformity as I’m not removing any tissue from the breast. In fact, the incision needed to accommodate the probe is always around 3 mm whether I treat a 1- or a 4-cm fibroadenoma. The patients have reduced stress because they can have the treatment in my office, and there’s no recovery time so they are immediately back to normal activities.
For more information: www.sanarus.com


 July 07, 2021
July 07, 2021