October 3, 2013 — Independent global research organization Frost & Sullivan honored Barco, a healthcare imaging solutions company, with the 2013 North American Product Line Strategy Award in Diagnostic Imaging Displays at the 2013 Growth, Innovation & Leadership Awards on Sept. 9, 2013 in San Jose, Calif. The award recognized Barco for delivering excellence and best practices in the North American diagnostic image displays market.
The Growth, Innovation and Leadership Awards are presented each year to companies that are predicted to encourage significant growth in their industries, have identified emerging trends before they became the standard in the marketplace and have created advanced technologies that will catalyze and transform industries in the near future.
Frost & Sullivan utilized the following criteria to benchmark Barco’s performance against key competitors: breadth of product line, size of addressable product base, impact on customer value, impact on market share and breadth of applications/markets served.
Empowering healthcare in an era of declining revenues
The research observed that, at this time of uncertainty in healthcare, providers that use the most efficient and widely applicable equipment are best positioned to face whatever challenges may lie ahead. In an era of declining reimbursements, revenue stability has become largely dependent upon a healthcare facility’s ability to increase productivity while maintaining high clinical standards.
High-value solutions enhance clinical productivity
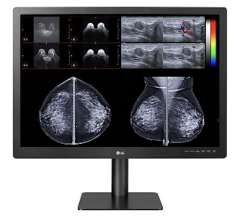
The organization lauded Barco’s Coronis Fusion 6MP display system and Mammo Tomosynthesis 5MP display system as two products that increase productivity and maintain the best possible image quality.
The Coronis Fusion 6 MP display system is the market’s first and only dual-head six megapixel color diagnostic display, which enables simultaneous viewing of two side-by-side 3 MP diagnostic images on a single monitor. An ongoing clinical study at Montefiore has demonstrated that the dual-head, bezel-less display system increases a reading physician’s productivity by 19 percent.
The market’s only diagnostic display designed and approved for breast tomosynthesis, the Barco Mammo Tomosynthesis 5 MP display system, is optimized to meet the quickly growing, critical need for breast imaging facilities across the nation.
For more information: www.frost.com

 March 12, 2024
March 12, 2024