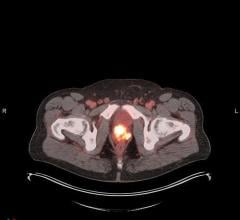
September 7, 2016 — Blue Earth Diagnostics announced that the first post-U.S. Food and Drug Administration (FDA) approval, commercial administrations of AxuminTM (fluciclovine F 18) injection occurred recently at Northside Hospital of Atlanta. Axumin is a novel molecular imaging agent indicated for use in positron emission tomography (PET) imaging in men with suspected prostate cancer recurrence based on elevated blood levels of prostate specific antigen (PSA) following prior treatment. It was recently approved by the FDA and is the first FDA-approved F-18 PET imaging agent indicated for use in patients with suspected recurrent prostate cancer.
“To date, we have had few imaging tools available for the evaluation of men with biochemically recurrent prostate cancer,” said William C. Lavely, M.D., nuclear medicine specialist, Northside Radiology Associates. “The approval of F-18 fluciclovine (Axumin) gives us an effective molecular imaging tool to evaluate these patients and assist clinicians in directing further management. Our initial experience is positive, demonstrating abnormal uptake in locations of potential metastatic prostate cancer. In my opinion, this prostate cancer PET agent, its clinical use for the evaluation of recurrent prostate cancer, and the additional information it provides for developing patient management plans has the potential to lead to better outcomes in men with recurrent prostate cancer. Further studies are necessary to determine these potential benefits.”
Axumin is commercially available through the national radiopharmacy network of Blue Earth Diagnostics’ exclusive U.S. commercial manufacturer and distributor, Siemens’ PETNET Solutions. Initial commercial production of Axumin is underway at certain regional radiopharmacies, and increasingly broader availability is planned in coming months.
Axumin was developed to enable visualization of the increased amino acid transport that occurs in many cancers, including prostate cancer. It consists of a synthetic amino acid that is preferentially taken up by prostate cancer cells compared with surrounding normal tissues, and is labeled with the radioisotope F18 for PET imaging.
For more information: www.blueearthdx.com

 July 30, 2024
July 30, 2024