April 23, 2015 — The U.S. Food and Drug Administration (FDA) has approved Siemens Healthcare’s 3-D mammography, breast tomosynthesis imaging system. This marks the 3-D mammography vendor to enter the U.S. market.
Siemens’ Mammomat Inspiration with Tomosynthesis Option is a breast tomosynthesis add-on option for Siemens Mammomat Inspiration digital mammography platform. Siemens’ breast tomosynthesis algorithm reconstructs multiple 2-D images of the breast into an approximation of a 3-D image to enable detection of tumors that are hidden by overlapping breast tissue, enabling more accurate diagnosis than standard 2-D digital mammography and reducing the number of false-positive findings. In a recent study involving 22 readers with a broad range of reading experience, Siemens demonstrated that all readers improved their accuracy in detecting and diagnosing cancers when reading digital breast tomosynthesis as an adjunct to full-field digital mammography.
In tomosynthesis mode, the X-ray tube of the Mammomat Inspiration digital mammography system rotates in a circular motion around the breast to acquire an image every two degrees while moving through an angular range of 50 degrees. The resulting 25 projections are reconstructed as three-dimensional digital breast tomosynthesis (DBT) images.
Conventional analog mammography and full-field digital mammography display only the 3-D structure of the breast on a 2-D level, hampering physicians’ efforts to identify certain types of tumors since anatomical structures in the breast can overlap and obscure lesions. Tomosynthesis acquires several breast projections from different angles and uses raw data to generate a 3-D volume dataset. Using this data set, clinicians can better analyze the type and size of breast lesions as well as microcalcifications compared to other forms of mammography. Breast tomosynthesis increases mammography’s sensitivity and specificity, in addition to improving efforts to differentiate and classify breast tumors.
“Our clinical data has demonstrated that the addition of Siemens’ digital breast tomosynthesis to a patient’s traditional 2-D digital mammogram increases detection of breast tumors. We know that in clinical practice, this increased diagnostic accuracy also means fewer diagnostic biopsy procedures and fewer anxiety-inducing recalls, which typically contributes to both improved patient outcomes and reduced cost,” said Gregory Sorensen, M.D., president and CEO of Siemens Healthcare North America. “With the FDA approval of the Mammomat Inspiration with Tomosynthesis Option, Siemens Healthcare reaffirms its longstanding commitment to cutting-edge innovation in women’s health.”
The Mammomat Inspiration with Tomosynthesis Option has been commercially available and used clinically for diagnosis since 2009 in Europe, Asia, and South America. The4 new breast tomosynthesis option is available on the company’s Mammomat Inspiration and Mammomat Inspiration Prime Edition digital mammography systems.


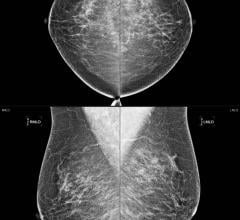
 July 29, 2024
July 29, 2024