October 2, 2007 — Confirma announced that it has installed CADstream for prostate at research sites including MRI at Belfair, Beth Israel, Simonmed and Lahey Clinic.
These research sites are assisting Confirma in the development and commercialization of CADstream for prostate. The prostate application will offer physicians comprehensive and clinically valuable tools for improved quality, standardization and efficiency in MRI studies of the prostate. The company recently exhibited at the National Conference on Prostate Cancer in Los Angeles and the AdMeTech Foundation Conference in Washington, DC with its research version of CADstream for prostate.
Prostate cancer is the second leading cause of cancer death in men. The need for prostate health management is growing rapidly, due to the increasing awareness that early detection will lead to long-term survival rates.
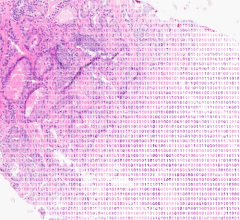
MR imaging is recognized as a sensitive tool for the noninvasive evaluation of prostate cancer. This emerging imaging modality is showing proven sensitivity for staging and determining extent of disease. Prostate MRI is a less invasive procedure using imaging, rather than biopsy first, and enables the physician to target areas of the body more precisely for biopsy planning.
For more information: www.confirma.com

 September 07, 2023
September 07, 2023