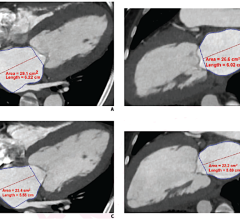
Image courtesy of AZE Ltd.
November 26, 2014 — AZE Technology announced the U.S. Food and Drug Administration (FDA) approval of Phoenix (Volume Registration Viewer), a viewer developed with a new concept for efficient and accurate image interpretation. Phoenix was designed with background volume processing that performs at the same time as data input, and presents various outcomes that were not possible with existing viewers.
Phoenix was approved against the previously cleared predicate device VirtualPlace (VP) from AZE, Ltd. Two sample images were used for this validation: a thoracic CT image and a thoracic MR image. Three observers made independent observations of 13 measurements with corresponding measurement tools in the two software programs (Phoenix and VirtualPlace) from the CT and MR images.
The only significant differences between Phoenix and the market-cleared VirtualPlace software measurement tools were RECIST measures. Additionally measurements with Phoenix tools on a phantom of known diameter came up within two percent of the known values.
Phoenix is designed for viewing CT/MRI images at an advanced level. Using fusion and superimposition or tumor size comparisons provides radiologists information faster. This technology is used in tumor evaluation for not only cancer patients, but for any type of mass being evaluated.
Instant matching of section positions helps reduce the time involved in reading exams, which increases the turnaround of clinical reports.
Phoenix is enhanced with multiple advance tools, hanging protocols and Smart Tags. AZE VirtualPlace 3-D workstation can also be interfaced with Phoenix.
For more information: www.azetech.com

 February 01, 2024
February 01, 2024