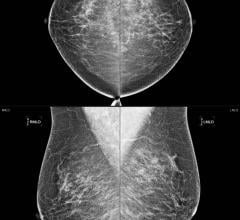
October 30, 2017 — The U.S. Food and Drug Administration (FDA) last week issued a release reminding patients that mammography is still the most effective breast cancer screening test. According to the agency, proper breast cancer screening lets healthcare providers check for cancer even before there may be signs and symptoms of the disease.
In the release, the FDA said it has received reports from healthcare providers and patients that some health centers are providing information that can mislead patients into believing that thermography — a type of test that shows patterns of heat and blood flow on or near the surface of the body — is a proven alternative to mammography. The agency said, however, it is not aware of any scientific evidence to support these claims.
Indeed, thermography has not been shown to be effective as a standalone test for either breast cancer screening or diagnosis in detecting early-stage breast cancer, the agency concluded.
“Plenty of evidence shows that mammography is still the most effective screening method for detecting breast cancer in its early, most treatable stages,” said Helen J. Barr, M.D., director of the Division of Mammography Quality Standards in the FDA’s Center for Devices and Radiological Health. “You should not rely solely on thermography for the screening or diagnosis of breast cancer.”
The FDA regulates the medical devices used for breast cancer screening. About 1 in 8 women will be diagnosed with breast cancer sometime in their lives, according to the National Cancer Institute, part of the National Institutes of Health. Rarely, men also can have breast cancer. But there has been a decline in breast cancer deaths in recent years, and one reason is because cancers have been detected earlier through mammography, according to the American Cancer Society.
In fact, according to the FDA, the greatest danger from thermography is that those who opt for this method instead of mammography may miss the chance to detect cancer at its earliest stage.
Thermography has only been cleared by the FDA as an adjunctive tool for use alongside a primary test like mammography. Patients who undergo a thermography test alone should not be reassured of the findings because the device was not cleared to be used other than with another testing method like mammography. The FDA provides more information about mammography for patients — including how it works and how to find a certified facility — on its website.
Moreover, some websites claim that thermography can find breast cancer years before it would be detected through other methods and have unproven claims about improved detection of cancer in dense breasts. The FDA is aware of no evidence that supports these claims, the release stated.
The FDA has taken regulatory action (including issuing warning letters) against healthcare providers and thermography manufacturers who try to mislead patients into believing that the thermography can take the place of mammography. To protect the public health, the FDA’s regulatory action can include scheduling a regulatory meeting, sending a warning letter or other correspondence, an establishment inspection, and judicial actions.
The FDA said it continues to monitor this situation.
Some women have sought out thermography because it is painless and does not require exposure to radiation. The FDA advises that if patients are worried about how a mammogram feels, they should talk to their healthcare provider about what they can expect. A mammogram can be uncomfortable for the person being screened because it briefly presses down on the breast to flatten out the breast tissue and increase the clarity of the X-ray image. The agency also has a section on its website about what patients can expect during a mammogram.
The agency also recommends patients should talk to their healthcare provider if they have specific questions about mammography, including questions about when and how frequently they should be screened. As a rule, patients should also call their healthcare provider if they notice any change in either of their breasts such as a lump, thickening or nipple leakage, or changes in how the nipple looks.
For more information: www.fda.gov

 July 29, 2024
July 29, 2024