June 13, 2008 - Codonics brought to SNM 2008, the Virtua Medical Disc Publisher, designed to set a new standard for ...
June 10, 2008 – The design of the University of Chicago Medical Center’s New Hospital Pavilion offers flexibility by ...
Researchers are using speckle tracking imaging in heart ultrasound to monitor the health of transplant acceptance, and ...
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
June 10, 2008 - The Virginia Commonwealth University Health System (VCU) recently implemented McKesson’s Horizon Medical ...
June 10, 2008 – MEDSEEK, a provider of enterprise portal management solutions, said today it is supporting Microsoft ...

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
June 10, 2008 - The use of ultrasound contrast agents during stress echocardiograms is safe, according to results ...
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
Could the treatment of Alzheimer’s disease one day become as routine as treating high cholesterol?
June 13, 2008 - The FDA granted tentative approval for Covidien’s Abbreviated New Drug Application (ANDA) for its Kit ...
June 10, 2008 - Press Ganey Associates Inc. today released “The 2008 Emergency Department Pulse Report: Patient ...
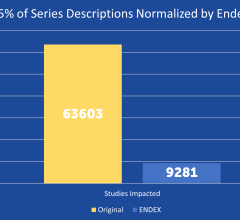
Did you know that approximately one-third of all the data in world is created by the healthcare industry and that ...
June 10, 2008 - At the Digestive Disease Week Conference, Cook Medical announced that it has been granted 510(k) clearance from the FDA for use of the Evolution Controlled Release Esophageal Stent System, a stent designed to improve the quality of life for patients with esophageal cancer.
Cook Medical's FDA-cleared Evolution Controlled Release Esophageal Stent System is a stent designed to improve the quality of life for patients with esophageal cancer.
June 10, 2008 - Wright Medical Group Inc., an orthopedic medical device company, said it has acquired certain assets of ...
Having the most efficient clinical workflows with enhanced diagnostic capabilities is a major goal for clinicians and ...
June 10, 2008 – Screening for lung cancer with computed tomography (CT) may help reduce lung cancer deaths in current ...
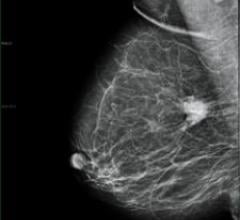
Candelis received 510(k) marketing clearance for its ImageGrid Mammography Web Viewer and ImageGrid Radiology Web Viewer ...
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
Acusphere has submitted a New Drug Application (NDA) to the FDA for approval to market Imagify (Perflubutane Polymer Microspheres for Injectable Suspension), an ultrasound imaging agent for the detection of coronary artery disease, which could prove as accurate as nuclear stress testing.
June 9, 2008 - Curlin Medical was recently awarded a three-year contract to provide its advanced pain management-local ...
June 9, 2008 - Hologic Inc. said today that it has signed a definitive agreement to acquire Third Wave Technologies Inc ...
Toshiba Teli America introduced the T24MSA001-MD medical-grade 24-inch LCD color widescreen monitor with full HD (1,080 ...
ProHance is a macrocyclic, nonionic, gadolinium-based magnetic resonance imaging (MRI) contrast agent designed for use in MRI in adults and children over two years of age to visualize lesions with abnormal vascularity in the brain (intracranial lesions), spine and associated tissues. ProHance is also indicated for use in MRI in adults to visualize lesions in the head and neck.
June 9, 2008 – Electronic health records maker MediNotes Corp. said it added 199 new software agreements with new and ...

 June 09, 2008
June 09, 2008