March 25, 2008 - Improved outcomes in patients with community-acquired pneumonia (CAP) led to top honors for AnMed ...
March 24, 2008 – AdvanDx received Frost and Sullivan’s 2008 Technology Innovation of the Year Award in the field of ...
March 24, 2008 - E-Z-EM Inc. stockholders voted in favor of a merger agreement providing for the acquisition of the ...
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
March 24, 2008 - Toshiba America Medical Systems installed its 500th Vantage 1.5T MR system at Jefferson Regional ...
March 24, 2008 - The American public believes doctors and physicians would do a good job running hospitals in their ...

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
March 24, 2008 - Midwestern State University awarded iCRco Inc. a multi-unit CR contract, and has already installed two ...
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
March 24, 2008 - Varian Medical Systems has formed a council of leading cancer centers in North America and Europe to ...
March 24, 2008 – Nucletron will be running a series of “Essentials in Brachytherapy” workshops across North American ...
March 24, 2008 – Siemens officially released the new Biograph 40 TruePoint PET-CT system and celebrated the first ...
Did you know that approximately one-third of all the data in world is created by the healthcare industry and that ...
March 20, 2008 – The number of CT procedures performed continues to grow despite a slow down in CT system purchases from ...
March 21, 2008 - Florida-based All Children's Hospital recently replaced its first round of mobile computing carts with ...
Kubtec featured its XPERT 40 digital specimen radiography device.
Having the most efficient clinical workflows with enhanced diagnostic capabilities is a major goal for clinicians and ...
March 21, 2008 - Cordis Corp. has received 510(k) marketing clearance from the FDA for the S.M.A.R.T. Nitinol Stent Transhepatic Biliary System for lengths of 120 mm and 150 mm, the company reported at the 33rd Annual Scientific Meeting of the Society of Interventional Radiology (SIR) meeting.
March 21, 2008 - Drawing on its experience in disease management for Medicaid patients and consumer-based technologies ...
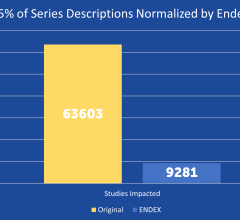
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
March 21, 2008 - The FDA gave conditional approval to PLC Systems to begin enrollment in a U.S. pivotal trial to study ...
March 21, 2008 - The Premier healthcare alliance said yesterday it is expanding its relationship with Alegent Health ...
March 20, 2008 - Harris Corp. was recently awarded a contract by the U.S. Department of Health and Human Services to ...
The Vivid S6 signature class cardiovascular ultrasound system reportedly combines strong performance and excellent image ...
March 20, 2008 – Siemens Medical Solutions received FDA 510(k) market clearance for the sale and distribution of two ...
March 20, 2008 - Welch Allyn recently announced at the 2008 HIMSS show that it created a new certification program for ...

 March 24, 2008
March 24, 2008