April 19, 2012 — GE Healthcare this week announced it has received U.S. Food and Drug Administration (FDA) clearance of Q.Freeze, one of the positron emission tomography/computed tomography (PET/CT) quantitative imaging technologies designed to enable treatment evaluation earlier in a patient’s cancer treatment.
April 19, 2012 — Carestream Healthcare announced it received U.S. Food and Drug Administration (FDA) clearance for its Carestream DRX-Revolution Mobile X-Ray System, and is now accepting orders from U.S. healthcare facilities.
April 19, 2012 - The FDA received a report from a hospital that 16 patients had developed hospital-acquired infections with the bacteria Pseudomonas aeruginosa following an examination with transesophageal echo (TEE) ultrasound probes using Other-Sonic Generic Ultrasound Transmission Gel. Upon investigation, theultrasound gel was found to be contaminated with the bacteria Pseudomonas aeruginosa and Klebsiella oxytoca. Manufactured by Pharmaceutical Innovations Inc., the non-sterile gel is used in ultrasound procedures to improve the transmission of the ultrasound signal from the transducer to the body.
Fujifilm’s APERTO Lucent is a 0.4T mid-field, open MRI system addressing today’s capability and image quality needs ...
April 17, 2012 -- Two hospitals announced treating their first patients with the Curve Image Guided Surgery system by Brainlab, which offers a command and control center for information-guided surgery.
April 17, 2012 - NoMoCo Inc.'s 12-piece pillow support system products optimize brain images for accurate, detailed diagnostic reports by the neuroradiologist in research centers, hospitals or clinical settings. The pillows are precision cut, designed from high-grade memory foam, easily cleanable and artifact free. NoMoCo helps patients complete radiologic exams with maximum comfort and with minimal movement, eliminating the need for repeat tests.

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
April 17, 2012 — Radius LLC is expanding its unique pay-per-study RIS/PACS model to include the option of on-demand 3-D imaging reconstruction.
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
April 17, 2012 - On April 9, 2012, the FDA's Center for Devices and Radiological Health (CDRH) launched its second version of the Innovation Pathway, called "Innovation Pathway 2.0." This program offers new and modified tools and methods to deepen collaboration between the FDA and innovators early in the process, prior to pre-market submission, with the goal of making the regulatory process more efficient and timely.
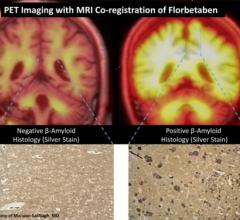
April 16, 2012 — Piramal Healthcare Ltd. has signed an agreement to acquire worldwide rights to the molecular imaging research and development portfolio of Bayer Pharma AG through its newly created subsidiary, Piramal Imaging SA. The portfolio includes rights to florbetaben, which is in the final stages of its Phase III clinical trials.
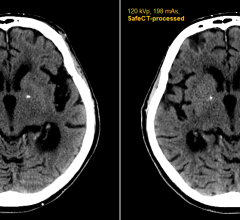
April 16, 2012 — Medic Vision Imaging Solutions Ltd. announced that within six months of clinical use in the United States, its SafeCT image enhancement system has delivered diagnostic image quality to more than 20,000 CT (computed tomography) studies acquired with low-dose protocols.
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
April 16, 2012 - Prime Healthcare Services (PHS), a hospital management company based in Ontario, Calif., currently owns and operates 16 acute-care hospitals. In February 2012, PHS signed a master agreement with Infinitt North America to replace PACS and cardiology PACS at several of its California hospitals.
April 16, 2012 — Philips Healthcare announced it is collaborating with Brainlab AG to create a comprehensive intra-operative magnetic resonance imaging (MRI) solution with the goal of streamlining neurosurgery procedures. Ingenia MR-OR is based on Philips’ digital broadband Ingenia MRI system, 1.5T and 3.0T, and is designed to be combined with Brainlab’s integrated operating room (OR) solutions.
April 16, 2012 — Elekta is revealing innovations for Leksell Gamma Knife Perfexion this week at the 2012 American Association of Neurological Surgeons (AANS) Annual Scientific Meeting in Miami.
Did you know that approximately one-third of all the data in world is created by the healthcare industry and that ...
April 13, 2012 — Leading experts in radiosurgery of the spine will present information about how to treat spinal tumors effectively using stereotactic radiosurgery rather than invasive surgical procedures during the annual meeting of the American Association of Neurological Surgeons (AANS) later this week in Miami.
April 13, 2012 — Novian Health, developer of Novilase Breast Therapy, announced it has begun a multicenter clinical trial on the use of Novilase Interstitial Laser Therapy (ILT) for the destruction of malignant breast tumors. The first facility to participate in the trial is the Rose Breast Center in Denver, under the study direction of Barbara Schwartzberg, M.D.
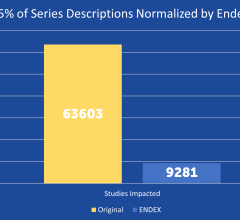
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
April 13, 2012 — The spiraling cost of silver is making medical films more and more obsolete, as new and more efficient medical imaging information systems (MIIS) continue to emerge, according to a new report by pharmaceutical and medical intelligence analysts GBI Research.
April 13, 2012 — Carestream is supporting a series of world-class mammography education, training and professional development opportunities, including continuing education (CE) and continuing medical education (CME) courses, to keep radiologists and technologists updated on the latest breast detection trends.
April 13, 2012 — Researchers at The Ohio State University (OSU) Wexner Medical Center have successfully used nanotechnology to target a protein that plays a key role in atherosclerosis and inflammation, and say the study is an important advance toward using immunotherapy to simultaneously diagnose and treat cardiovascular disease.
April 13, 2012 — GE Healthcare announced the preliminary results of two Phase 3 studies of its investigational positron emission tomography (PET) amyloid imaging agent, F-18 flutemetamol, where both studies met their primary endpoints.
April 12, 2012 — U-Systems announced that the U.S. Food and Drug Administration’s (FDA) Radiological Devices Panel of the Medical Devices Advisory Committee recommended approval of the somo•v Automated Breast Ultrasound (ABUS) system.
April 12, 2012 — Several innovative cardiovascular and radiology technologies were featured in Medical Device and Diagnostic Industry (MD+DI) magazine’s April issue, which also announced the finalists in the 2012 Medical Design Excellence Awards (MDEA) competition.

 April 19, 2012
April 19, 2012