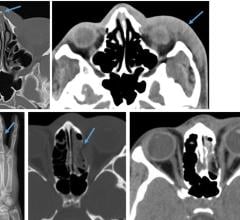
January 11, 2018 — Researchers using functional magnetic resonance imaging (fMRI) have found that neurofeedback training ...
The Centers for Medicare & Medicaid Services (CMS) Center for Medicare and Medicaid Innovation (Innovation Center) announced the launch of a new voluntary bundled payment model called Bundled Payments for Care Improvement Advanced (BPCI Advanced). Under traditional fee-for-service payment, Medicare pays providers for each individual service they perform. Under this bundled payment model, participants can earn additional payment if all expenditures for a beneficiary’s episode of care are under a spending target that factors in quality.
Exhibitors at the 2017 Radiological Society of North America (RSNA) meeting rode the artificial intelligence (AI) ...
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
Competitive male triathletes face a higher risk of a potentially harmful heart condition called myocardial fibrosis, according to research presented at the annual meeting of the Radiological Society of North America (RSNA), Nov. 26-Dec. 1 in Chicago. The increased risk, which was not evident in female triathletes, was directly associated with the athletes’ amount of exercise.
January 9, 2018 – The most common type of tumor found in the kidney is generally quite small (less than 1.5 in). These ...

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
Konica Minolta President of Healthcare IT Steve Deaton details new features in the Exa Platform designed to improve ...
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
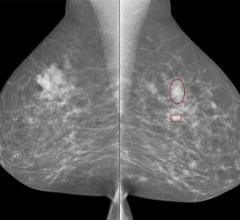
Women prefer to get their mammograms every year, instead of every two years, according to a new study presented at the annual meeting of the Radiological Society of North America (RSNA), Nov. 26-Dec. 1 in Chicago.
School-age football players with a history of concussion and high impact exposure undergo brain changes after one season of play, according to two new studies conducted at UT Southwestern Medical Center in Dallas and Wake Forest Baptist Medical Center in Winston-Salem and presented at the 2017 annual meeting of the Radiological Society of North America (RSNA), Nov. 26-Dec. 1 in Chicago.
Radiologic signs of injury could help identify victims of intimate partner violence, according to a study presented at the annual meeting of the Radiological Society of North America (RSNA), Nov. 26-Dec. 1 in Chicago.
Did you know that approximately one-third of all the data in world is created by the healthcare industry and that ...
January 8, 2018 — EchoPixel showcased the latest version of True 3D, its interactive, mixed reality software solution at ...
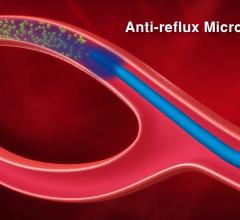
Guerbet announced that it has entered into an agreement under which it will acquire Israeli company Accurate Medical Therapeutics , which specializes in the development of microcatheters used in interventional radiology.
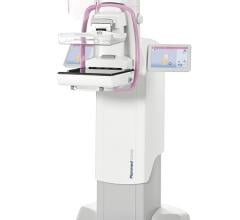
The U.S. Food and Drug Administration (FDA) issued an approval letter for the Planmed Clarity 2-D full-field digital mammography (FFDM) system on Dec. 28, 2017, the company announced. The Planmed Clarity 2-D system is a digital mammography system that provides high image quality, optimal ergonomics and excellent usability for breast cancer screenings, according to the company. In addition to screening mammograms, the system supports diagnostic work-up examinations and biopsy procedures.
Having the most efficient clinical workflows with enhanced diagnostic capabilities is a major goal for clinicians and ...
Here is the list of the most popular articles and videos on the Imaging Technology News (ITN) magazine website from the ...
The webinar "Neuroimaging from a Clinical MRI Perspective," explains how imaging departments can become more creative to ...
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
Canon Inc. and Canon Medical Systems Corp. announced the official corporate name (trade name) change of Canon Group Company Toshiba Medical Systems Corp. to Canon Medical Systems Corp. (hereafter referred to as Canon Medical Systems).
Women with higher body mass index (BMI) face an increased risk of not detecting their breast tumor until it has become large, according to a study presented at the 2017 annual meeting of the Radiological Society of North America (RSNA) in Chicago in November. Researchers said the findings suggest that women with higher BMI may need shorter intervals between mammography screening exams.
Mevion Medical Systems has received U.S. Food and Drug Administration (FDA) 510(k) clearance for the Mevion S250i Proton Therapy System including Hyperscan pencil beam scanning (PBS) technology. Hyperscan PBS introduces novel energy layer switching and automated collimation systems. These advantages enable the S250i system to deliver faster, sharper and more robust PBS proton radiation treatments.
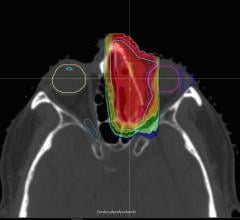
Although targeted therapies have produced dramatic advances in the ability to control some types of advanced lung cancer, growth of the disease in the brain remains a major problem. Radiation is often used to treat deposits in the brain, but the best technique to deliver radiation can be controversial. Whole-brain radiation therapy, as its name suggest, treats the entire brain but can be associated with notable cognitive side effects. Another strategy, radiosurgery, directs highly-focused radiation only to the sites of metastasis, largely sparing the normal brain.
January 3, 2018 — medQ Inc. recently announced the launch of its Q/ris MIPS Reporting module. This module is designed to ...
The U.S. Food and Drug Administration (FDA) issued the final version of the guidance, “Technical Considerations for Additive Manufactured Medical Devices." Additive manufacturing (AM), the broad category of manufacturing encompassing 3-dimensional (3-D) printing, is an emerging technology. This guidance is not intended to introduce new policy, but rather outlines the Agency’s current thinking about the technical aspects associated with AM processes, and provides manufacturers with recommendations for device design, manufacturing and testing considerations for use when developing devices that include at least one additively manufactured component or additively fabricated step.

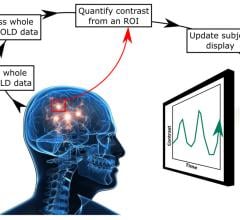
 January 11, 2018
January 11, 2018