August 13, 2012 — Bayland Digital Inc. is a free medical scanning service that converts film X-rays into digital format and then recycles the old X-rays.
August 13, 2012 — To improve magnetic resonance (MR) exam efficiency and image quality, Toshiba America Medical Systems Inc. has received U.S. Food and Drug Administration (FDA) clearance for its high-density 16-element flexible coil system, developed in partnership with NeoCoil. The new coil system makes it easier for clinicians to complete high-quality exams and improve diagnostic efficiency.
August 13, 2012 — Royal Philips Electronics and Celsion Corp. announced that the U.S. Food and Drug Administration (FDA) has provided clearance to initiate a clinical study supporting a joint development program for Celsion’s ThermoDox combined with Philips' Sonalleve MR-HIFU (magnetic resonance-guided high intensity focused ultrasound) technology for the palliation of painful metastases to the bone caused by lung, prostate or breast cancers.
Fujifilm’s APERTO Lucent is a 0.4T mid-field, open MRI system addressing today’s capability and image quality needs ...
August 13, 2012 — The new Harvey L. Neiman Health Policy Institute (HPI), established by the American College of Radiology (ACR), will examine the role of radiology in new healthcare delivery and payment models — particularly quality-based approaches to radiologic care and the impact of medical imaging on overall healthcare costs.
Aug. 8, 2012 — iHeart Centers acquired the Heart IT WebPAX system for facilitating the management of medical images. The zero-footprint medical image workstation provides Web-based medical image management technology and services to healthcare systems, large hospitals and private clinics.

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
August 7, 2012 — RadNet Inc. announced it will offer Amyvid, a radioactive tracer used in positron emission tomography/computed tomography (PET/CT) imaging, at more locations across the United States. Amyvid is the first test that can effectively rule out Alzheimer's disease (AD) in patients who are being evaluated for cognitive impairment.
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
August 7, 2012 — Navidea Biopharmaceuticals Inc., a company focused on precision diagnostic radiopharmaceuticals, announced it has entered into an agreement with Alseres Pharmaceuticals Inc. to license [123I]-E-IACFT Injection (CFT), an iodine-123 radio-labeled imaging agent being developed as an aid in the diagnosis of Parkinson’s disease and other movement disorders, with a potential use as a diagnostic aid in dementia.
August 7, 2012 —Brainlab and Elekta have announced the first installation of an Elekta linear accelerator (linac) equipped with the ExacTrac patient positioning system by Brainlab; a strategy to make radiosurgery treatments for cancer patients more accurate and comfortable.
August 7, 2012 — ClearCanvas Inc. announced the release of ShareAgent, a routing application that allows healthcare professionals to transfer medical images to any DICOM (digital imaging and communications in medicine)-enabled device, and ShareStation, a mini-PACS (picture archiving and communications system) solution.
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
August 7, 2012 — Fujifilm Medical Systems U.S.A. announced the latest addition to its family of digital radiography (DR) products, the FDR D-EVO Suite II, a cost-effective, flexible, DR-room solution designed to optimize workflow while utilizing any of Fujifilm’s FDR D-EVO portable detectors.
August 7, 2012 — A new method of computed tomography (CT) could produce significantly improved images of knee, spine and hip implants, and may lower radiation exposure, suggested preliminary research presented at the 2012 American Association of Physicists in Medicine (AAPM) annual meeting.
August 7, 2012 — A new type of mammography could accurately measure breast density, one of the most significant risk factors for breast cancer, suggested early research presented at the 2012 American Association of Physicists in Medicine (AAPM) annual meeting.
Did you know that approximately one-third of all the data in world is created by the healthcare industry and that ...
August 6, 2012 — In an effort to provide small hospitals and practices an economical path from analog to digital radiology, Agfa launched the CR-10-X computed radiography (CR) imaging digitizer.
August 6, 2012 — PACSGear, a provider of imaging connectivity for electronic health records (EHR), announced an agreement with eMix, a cloud-based virtualized electronic medical information exchange, to participate in the Open Image Exchange, a cloud-based network designed to securely share medical images and information.
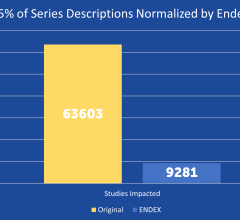
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
August 6, 2012 — MIM Software Inc. introduces ART Assist, an automated processing and reporting tool designed to automatically quantify changes to a patient over the course of radiation delivery.
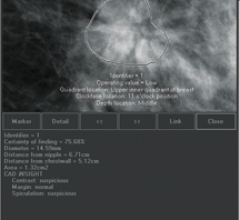
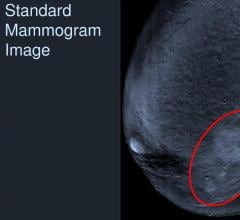
August 6, 2012 — iCAD Inc., a provider of advanced image analysis, workflow solutions and radiation therapy for the early identification and treatment of cancer, announced the availability of the PowerLook advanced mammography platform (AMP), its next-generation mammography computer-aided detection (CAD) system.
August 6, 2012 — Brachytherapy offers an effective and relatively cheap method of treating internal cancers, and its popularity in the future will likely rely on its financial status, according to a new report by GlobalData.
August 3, 2012 — Rolling Oaks Radiology, a division of RadNet Inc., announced it now offers GE Healthcare SenoBright contrast-enhanced spectral mammography (CESM) at their Oxnard, Calif., location.
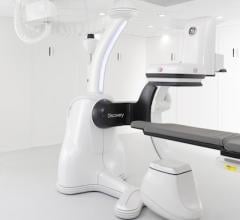
August 3, 2012 — GE Healthcare announced it has already received six orders for its Discovery IGS 730 system since receiving U.S. Food and Drug Administration (FDA) clearance in February 2012.
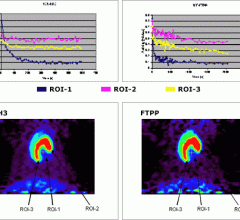
August 3, 2012 — FluoroPharma Medical Inc. announced that they have received high-quality images in an investigator-sponsored clinical trial in China where patients with coronary artery disease (CAD) were given BFPET, its imaging agent for measuring cardiovascular blood flow.

 August 13, 2012
August 13, 2012