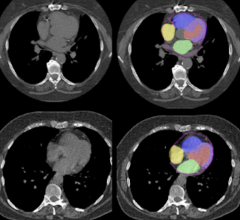
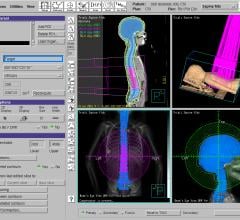
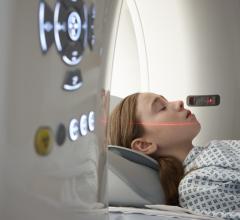
The FDA has granted clearance for the new series of CT systems that now include much of the advanced technology available on GE’s volume CT system, the LightSpeed VCT, in a smaller package. The new BrightSpeed scanners provide the same image quality and productivity capabilities of the LightSpeed systems, but on the new BrightSpeed 4-, 8- and 16-slice CT systems. The scanners can be installed in a similar footprint to a single-slice CT system, eliminating the need to renovate or enlarge existing radiology departments.


 May 29, 2024
May 29, 2024