May 25, 2023 — GE HealthCare, a leader in breast cancer care technology and diagnostics, has today announced that NCCN Guidelines for clinicians and patients now recommend the use of FES PET for ER+ positive disease under certain circumstances during the systemic staging workup of patients with recurrent or metastatic breast cancer. GE HealthCare’s Cerianna, available in the U.S., is the only FDA approved FES PET imaging agent. This inclusion in the NCCN Guidelines comes after the Society of Nuclear Medicine and Molecular Imaging (SNMMI) recently published Appropriate Use Criteria to guide referring and imaging physicians in appropriate use of estrogen receptor (ER)-targeted PET imaging with 16α-18F-fluoro-17ß Fluoroestradiol.
In the U.S., it is estimated that 181,0002 people have metastatic breast cancer (also called stage 4)3, with a five-year survival rate of 28 percent4. Currently, when treating metastatic breast cancer patients, oncologists base clinical decisions on diagnostic tools including biopsy results which only represent the sampled area of the tumor. However, ER expression – one of the most common breast cancer biomarkers - can vary both within the primary tumor, across different lesions5, and change over time.
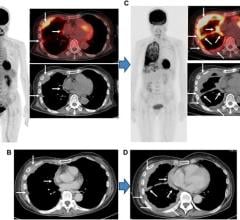
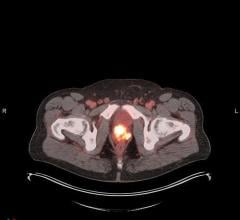
Cerianna is indicated for use with PET imaging for the detection of ER+ lesions as an adjunct to biopsy in patients with recurrent or metastatic breast cancer. Providing a whole-body view of ER+ lesions, Cerianna may deliver a comprehensive assessment to assist in making an informed diagnosis and treatment plan for the patient. This could potentially enable more targeted and individualized treatment strategy and avoid the selection of inappropriate or less effective therapies.
“The latest NCCN Guidelines reflect the impact the Cerianna PET is starting to have in the care of patients with metastatic breast cancer,” Dr. David Mankoff, Professor and Vice Chair of Research in radiology at University of Pennsylvania, commented. “As was the case for fluorodeoxyglucose (FDG) PET, inclusion in the NCCN guidelines will help support access for Cerianna PET for patients with MBC and provides impetus for the medical community to define the clinical scenarios where Cerianna PET will be most helpful.”
“FES PET, like FDG PET, is now included in the NCCN Guidelines. We have an opportunity to use FES PET to assess ER function in all tumor sites in patients with ER+ metastatic breast cancer,” Dr. Hannah M Linden, Breast Medical Oncologist, University of Washington and Fred Hutchinson Cancer Center in Seattle, Washington, said. “This is a helpful tool for diagnostic confirmation and may have the ability to aid in prognosis and prediction of clinical benefit from endocrine based therapies, including with CDK 4/6 inhibitors. We have many endocrine options now, and FES PET may identify patients who remain ER+ and thus potentially benefit from endocrine based therapy.”
“We are delighted to see the inclusion of Cerianna PET in the NCCN breast oncology guidelines, for potential use when oncologists are evaluating metastatic breast cancer patients,” Dr. Mark Hibberd, Chief Medical Officer at GE HealthCare’s Pharmaceutical Diagnostics segment, said. “Detecting ER+ lesions in patients with recurrent or metastatic breast cancer could potentially aid oncologists, surgeons and clinicians in choosing the most appropriate therapy for patients.”
The NCCN is a not-for-profit alliance of 33 leading U.S. cancer centers devoted to patient care, research, and education. The NCCN Guidelines are the recognized standard for clinical direction and policy in cancer care and are the most thorough and frequently updated clinical practice guidelines available in any area of medicine.
GE HealthCare’s Pharmaceutical Diagnostics segment is a global leader in imaging agents used to support around 100 million procedures per year globally, equivalent to three patient procedures every second. Its Molecular Imaging portfolio combines established proprietary products across cardiology, neurology and oncology, with an innovative pipeline, all aimed at enabling better informed diagnosis and monitoring for improved therapy decision making and clinical outcomes.
NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
For more information: www.gehealthcare.com
References:
1 J Nucl Med 2023 Mar;64(3):351-354. doi: 10.2967/jnumed.123.265420
2 Gogate A, et al. Projecting the Prevalence and Costs of Metastatic Breast Cancer From 2015 through 2030. JNCI Cancer Spectrum. August 2021
3 Mariotto et al, 2017
4 https://www.cancer.org/cancer/breast-cancer/understanding-a-breast-cancer-diagnosis/breast-cancer-survival-rates.html
5 Kurland, et al. Between-patient and within-patient (site-to-site) variability in estrogen receptor binding, measured in vivo by 18F-fluoroestradiol PET. J Nucl Med. 2011;52(10):1541-1549 / Currin, et al. Temporal Heterogeneity of Estrogen Receptor Expression in Bone-Dominant Breast Cancer: 18F-Fluoroestradiol PET Imaging Shows Return of ER Expression. J Natl Compr Canc Netw. 2016;14(2):144-147
Related FES PET Imaging Content:
GE Healthcare Advances Precision Health & Theranostics With New Products & Solutions
GE Healthcare Acquires Zionexa
PETNET Solutions and Zionexa USA Announce FDA Approval of Cerianna
Newly Approved PET Imaging Agent Solves Clinical Dilemmas in Breast Cancer Patients

 July 30, 2024
July 30, 2024