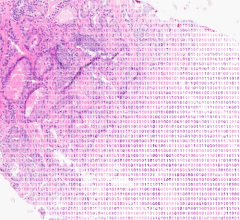
November 1, 2007 - The American College of Obstetricians and Gynecologists (ACOG) announced new recommendations emphasizing colonoscopy as the preferred method for colorectal cancer screening for both average-risk and high-risk women.
ACOG changed its colorectal cancer screening recommendations because colonoscopy allows for direct visualization of the entire colon surface, and it allows for removal of any precancerous polyps at the same time. Additionally, colonoscopy allows access to right-sided lesions-the main type of advanced colorectal cancer occurring in women-which are more likely to be missed by other screening methods. Previously, ACOG recommended that all women age 50 and older be screened for colorectal cancer by one of several methods, with no single method being preferred over another.
"Our message today is that colonoscopy is the gold standard when it comes to colorectal cancer screening. While we want ob-gyns to encourage this method, they should still discuss the advantages and limitations of the other screening options with their patients," said Carol L. Brown, M.D., immediate past vice chair of ACOG's Committee on Gynecologic Practice, which developed the recommendations. "The bottom line is we want women to get tested by whichever method they are most likely to accept and follow through with."
According to the Committee Opinion, ACOG does not recommend fecal DNA testing or computed tomography colonography (also known as "virtual colonoscopy") for screening outside the research setting, pending further data on their effectiveness.
For more information: www.acog.com


 November 11, 2025
November 11, 2025