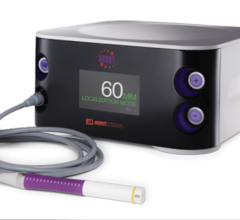
March 4, 2009 - Xoft Inc. said today the FDA cleared a skin and surface treatment applicator for use with the Axxent Electronic Brachytherapy (eBx) System to deliver surface brachytherapy, including Intraoperative Radiation Therapy (IORT).
This latest clearance allows the applicator to be used on any external or internal surface of the body where radiation therapy is indicated.
Previously cleared for accelerated treatment of early stage breast cancer and endometrial and rectal cancers, the Axxent System is also cleared for use in the treatment of surface cancers or conditions where radiation therapy is indicated. The proprietary Electronic Brachytherapy treatment platform is designed to deliver localized, non-radioactive, isotope-free radiation treatment in minimally-shielded clinical settings under the supervision of a radiation oncologist.
"As market and clinical adoption for Electronic Brachytherapy grows for our breast and endometrial applications, the FDA clearance of skin and surface indications completely supports Xoft's mission of expanding the number of patients who can access radiation therapy and can dramatically benefit from it," said Michael Klein, president and CEO of Xoft. "Not only does it create access to a fast, convenient, non-isotope treatment option for millions of skin cancer patients, it continues to expand the clinical utility of the broad based Electronic Brachytherapy treatment platform that is being used at more than 50 radiation oncology centers across the U.S."
Skin cancer is the most common cancer in the U.S. with more than 1 million cases diagnosed annually. If diagnosed and treated early, most cases of skin cancer can be cured; however the National Cancer Institute estimates that skin cancer results in more than 10,000 deaths each year. Radiation therapy is one of several primary treatment options used to treat skin cancers, including surgery and chemotherapy. Designed to kill cancer cells and reduce the spread and local recurrence of cancer, radiation therapy is used alone or in combination with surgery or chemotherapy, depending on the location and stage of the cancer tumor as well as the patient's general health.
In addition to skin indications, the FDA clearance also covers surface indications, which means that many intraoperative radiation therapy procedures can be performed with the Xoft source, while surgery is being performed. Intraoperative treatment potentially reduces the time required for radiation therapy by delivering it immediately during surgery before any remaining cancer cells have a chance to grow. IORT, utilizing the Axxent System's miniaturized X-ray source that can deliver localized and targeted radiation treatment, is designed to minimize radiation exposure to surrounding healthy tissue and enables physicians and treating professionals to remain in the operating room with the patient.
For more information: www.xoftinc.com

 July 11, 2024
July 11, 2024