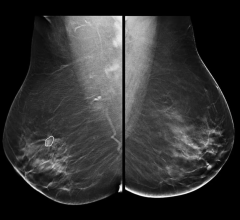
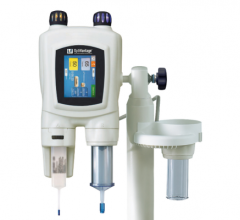
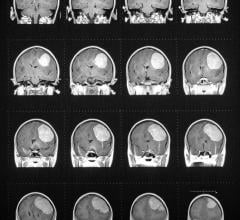
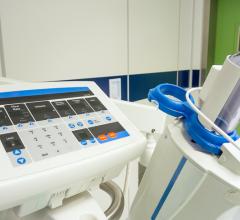
This convenient, point-of-care (POC), handheld device analyzes a finger prick sample of blood for creatinine levels—an important indicator of a patient’s renal function. The test can be performed in the imaging suite, with results available in as little as 30 seconds. Evaluating a patient’s kidney function can help determine their ability to safely receive IV contrast prior to CT and MR imaging—an important precaution especially for patients with risk factors such as diabetes, known kidney impairment, and advanced age. In addition, EZ Chem also features GFR result calculation and display.
October 2007


 April 12, 2024
April 12, 2024