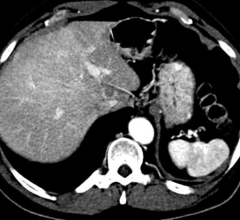
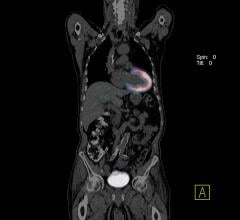
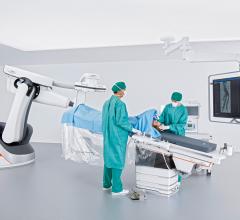
Central Minnesota residents now have access to advanced computed tomography (CT) technology that is safe and fast at St. Cloud Hospital with the installation of the Aquilion One/Genesis Edition from Toshiba Medical, a Canon Group company. The system is located across a hallway from the emergency department and is being used for a wide range of exams, including neuro perfusion studies.
The Medical Device Innovation, Safety and Security Consortium (MDISS) recently launched the first of more than a dozen planned device security testing labs and cyber-ranges. The new MDISS World Health Information Security Testing Lab (WHISTL) facilities will comprise a federated network of medical device security testing labs, independently owned and operated by MDISS-member organizations including healthcare delivery organizations, medical device manufacturers, universities and technology companies. Each WHISTL facility will launch and operate under a shared set of standard operating procedures. The goal is to help organizations work together to more effectively address the public health challenges arising from cybersecurity issues emergent in complex, multi-vendor networks of medical devices.
Insightec announced recently that worldwide adoption of magnetic resonance (MR)-guided focused ultrasound continues to gain momentum as Exablate Neuro patient treatment numbers surpass the 1,000 mark. Medical facilities are ramping up their MR-guided focused ultrasound service and essential tremor patients are now being treated on a weekly basis at many sites, according to the company.
Fujifilm’s APERTO Lucent is a 0.4T mid-field, open MRI system addressing today’s capability and image quality needs ...
A new UCLA study is the first to identify patient and tumor characteristics that predict the successful creation of models and which types of sarcomas are most likely to grow as xenografts. The research, which is the first and largest patient-derived orthopedic xenograft (PDOX) study in sarcoma, gives physician-scientists a much-needed roadmap for personalizing therapies for the disease without placing patients at risk for treatment-related complications or ineffective therapy.
Melissa Martin, MS, president of the American Association of Physicists in Medicine (AAPM), discusses her vision of the ...

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
When to initiate screening for breast cancer, how often to screen, and how long to screen are questions that continue to spark emotional debates. A new study compares the number of deaths that might be prevented as a result of three of the most widely discussed recommendations for screening mammography. Published early online in Cancer, a peer-reviewed journal of the American Cancer Society, the findings may provide valuable guidance to women and their physicians about choosing a screening regimen.
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
Jared Houk, vice president, imaging business unit, for Agfa HealthCare North America, previews the DR800 at AHRA 2017.
The U.S. Department of Health and Human Services (HHS), Office for Civil Rights (OCR) recently launched a revised web tool that puts important information on cybersecurity breaches into the hands of individuals. The tool is designed to empower them to better identify recent breaches of health information and to learn how all breaches of health information are investigated and successfully resolved.
Mark Pankuch, Ph.D., director of medical physics at the Northwestern Medicine Chicago Proton Center, discusses the ...
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
Accuray Inc. and the Summit Cancer Center-Boise announced that the center is now treating patients with the Radixact Treatment Delivery System, the next-generation TomoTherapy platform. The Radixact System replaced a competitive conventional linear accelerator in the single-vault freestanding facility.
August 16, 2017 — A new imaging information technology (IT) market report from Signify Research suggests enterprise ...
August 18, 2017 — SuperSonic Imagine recently introduced the newest version of its premium ultrasound system, Aixplorer ...
Did you know that approximately one-third of all the data in world is created by the healthcare industry and that ...
August 18, 2017 — The American Society of Nuclear Cardiology (ASNC) has released a joint expert consensus document with ...
August 17, 2017 — The proton center at MedStar Georgetown University Hospital will utilize RayStation for planning on ...
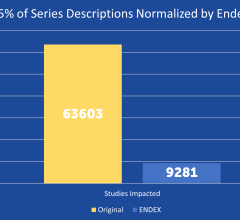
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
August 17, 2017 — Client Outlook recently announced that MaineHealth has selected eUnity as its enterprise image viewing ...
Pentax Medical Co. announced the U.S. launch of the Slim Linear Ultrasound Endoscope (EG-3270UK), the company's newest ultrasound endoscope.
Houston Methodist Hospital and Siemens Healthineers have entered into a multi-year agreement to bring cutting-edge technology to Houston Methodist that is not currently available in the Texas Medical Center. This agreement will provide innovative, world-class medical technology to Houston Methodist Hospital and all of its community hospital facilities.
It's critical for today's healthcare professionals to understand the balance between the risks and benefits of any X-ray imaging procedure.
ScImage Inc. was recently awarded a new DIN-PACS IV (Digital Imaging Network/Picture Archiving and Communications System) contract by the U.S. government. The contract is potentially valued up to $400 million, and the terms include one five-year base period and one five-year option period.
Carestream Health recently introduced new MR (magnetic resonance) Brain Perfusion and MR Brain Diffusion modules that enable radiologists to read these imaging studies from a Carestream Vue PACS (picture archiving and communication system) diagnostic viewer. The company has received U.S. Food and Drug Administration (FDA) 510(k) clearance for use in the United States, and these new tools also have been approved for use in Europe and other countries across the globe.


 August 23, 2017
August 23, 2017