August 29, 2007 - Results of the first large scale study comparing the results of a particular digital mammography ...
August 29, 2007 – Aurora Imaging Technology Inc. will serve as a flagship sponsor of the American Cancer Society’s ...
August 29, 2007 — EndoGastric Solutions, a leader in natural orifice surgery (NOS), announced it has completed a $30 ...
While most women understand the importance of health screenings, an estimated 72 million have missed or postponed a ...
August 29, 2007 — Performance management solutions company SciHealth was named to the first-ever Inc. 5000 magazine list ...
August 29, 2007 - Calgary Scientific Inc. Medical Group (CSIMG), a provider of advanced visualization and analysis ...

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
August 29, 2007 — InnerSpace Corp. announced it expanded its line of Datel brand organized storage cabinets to include ...
Fujifilm’s APERTO Lucent is a 0.4T mid-field, open MRI system addressing today’s capability and image quality needs ...
August 29, 2007 — Roche Diagnostics said the U.S. Food and Drug Administration approved its biologics license for a test ...
August 28, 2007 — To support initiatives that enhance patient safety and quality of care, Cardinal Health announced it ...
August 27, 2007 – Toshiba America Medical Systems announced the installation of two Infinix CF-i/BP vascular X-ray ...
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
August 28, 2007 – Arizona State University (ASU) recently announced that they have received the National Science ...
August 28, 2007 - Aurora Imaging Technology Inc. recently announced that the company is a flagship sponsor of the ...
August 28, 2007 — Biometric technology research and development firm M2SYS Technology said it has partnered with Gnosis ...
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
August 28, 2007 - Cedara Software, a Merge Healthcare company and a medical imaging software and services company ...
August 28, 2007 – GE Healthcare announced that it has signed an agreement with Novation for GE’s advanced radiology and ...
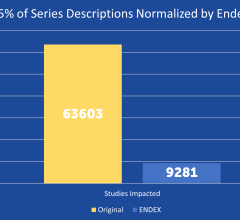
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
August 28, 2007 - Misys Healthcare Systems and iMedica jointly announced an agreement that Misys will license iMedica’s ...
August 28, 2007 - Clinical software maker Allscripts announced Southwestern Medical Clinic selected Allscripts as its ...
August 28, 2007 - Carestream Health, Inc. announced that Médical Tronik Montreal Ltd. has become the exclusive supplier ...
August 28, 2007 – CardiArc Inc. announced that researchers at Emory University Hospital will perform evaluations in ...
August 27, 2007 — iCRco Inc. a manufacturer of computed radiography and X-Ray Digitizing equipment recently gathered all ...
August 27, 2007 — CBaySystems and Services Inc., a leading global provider of transcription, BPO services and technology ...

 August 28, 2007
August 28, 2007 

