Toshiba Medical, a Canon Group company, introduced new educational tools and interactive learning resources to help healthcare providers stay current on CMEs and clinical education without impacting busy schedules. The new advancements give clinicians more accessible and mobile-friendly resources and accreditation opportunities that fit into their on-the-go schedules.
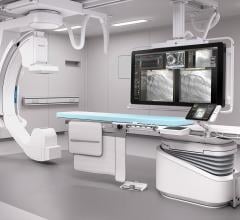
Philips recently announced the results of a comprehensive, independent, two-year study demonstrating the clinical workflow benefits of its next-generation image-guided therapy platform, Azurion. The study investigated nearly 800 patient procedures to evaluate the impact of Azurion at St. Antonius Hospital in Nieuwegein, the Netherlands. The data demonstrated clinicians’ use of Azurion resulted in significant time savings for the hospital, including a 17 percent reduction of the average interventional procedure time, a 12 percent reduction of in-lab patient preparation time, and a 28 percent reduction of post-procedure lab time.
Materialise and Formlabs are collaborating to deliver a complete, cost-effective and easy-to-use solution for hospitals looking to start an in-house 3-D print lab. The new offering combines the Materialise Mimics inPrint medical imaging software with Formlabs’ Form 2 professional-grade desktop 3-D printers. The components of this complete 3-D printing package facilitate the implementation of patient-specific care at an affordable price-per-print for quick return on investment.
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
Vital Images unveiled the newest version of Vitrea Advanced Visualization software, the cornerstone of its imaging platform, at the 2017 Radiological Society of North America (RSNA) annual meeting, Nov. 26-Dec. 1 in Chicago. The new release, dubbed Version 7, includes numerous product enhancements and state-of-the-art modality applications.
December 4, 2017 — Change Healthcare and Zebra Medical Vision announced an agreement to create solutions that deliver ...

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
The importance of quantitative analysis in breast imaging was the focus of numerous abstracts accepted for presentation at the 103rd Annual Radiological Society of North America (RSNA) meeting, Nov. 26-Dec. 1 in Chicago. Four abstracts highlighted the use of Volpara Solutions' quantitative analysis tools for breast imaging. The tools provide actionable quantitative metrics for clinicians, including volumetric density, personalized dose, potential high risk, and other factors designed to maintain accuracy and drive consistent quality in breast screening.
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
ScImage Inc. introduced PicomPassport at the Radiological Society of North America’s 103rd Annual Scientific Assembly, Nov. 26-Dec. 1 in Chicago. PicomPassport is a new technology that leverages PICOM365’s Azure-based Cloud to simplify the importing of external patient exams into any picture archiving and communication system (PACS).
November 29, 2017 — Philips announced the introduction of IntelliSpace Enterprise Edition for Radiology, its ...
November 28, 2017 — Hologic Inc. announced it has signed a development and distribution agreement with Clarius Mobile ...
Did you know that approximately one-third of all the data in world is created by the healthcare industry and that ...
November 28, 2017 — Fujifilm Medical Systems U.S.A. Inc. displayed the all-new FDR Go Plus version portable digital ...
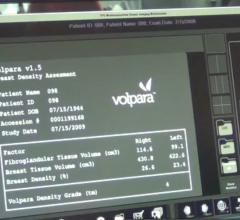
Hologic Inc. announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Quantra 2.2 Breast Density Assessment Software. The software enables clinicians to provide women with consistent breast density assessments during routine breast cancer screenings.Hologic Inc. announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Quantra 2.2 Breast Density Assessment Software. The software enables clinicians to provide women with consistent breast density assessments during routine breast cancer screenings.Hologic Inc. announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Quantra 2.2 Breast Density Assessment Software. The software enables clinicians to provide women with consistent breast density assessments during routine breast cancer screenings. Hologic Inc. announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Quantra 2.2 Breast Density Assessment Software. The software enables clinicians to provide women with consistent breast density assessments during routine breast cancer screenings.
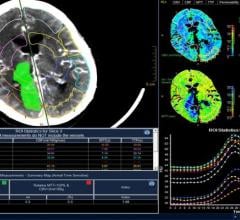
Infervision is introducing what it calls the first and only artificial intelligence (AI) platform to help radiologists detect and diagnose stroke faster, leading to patients getting life-saving treatment when time is of the essence. This new stroke detection solution is being introduced at the 2017 Radiological Society of North America (RSNA) conference, Nov. 26-Dec. 1 in Chicago.
Having the most efficient clinical workflows with enhanced diagnostic capabilities is a major goal for clinicians and ...
November 27, 2017 — At the 103rd Scientific Assembly and Annual Meeting of the Radiological Society of North America ...
November 27, 2017 — Artificial intelligence (AI) startup Aidoc announced that it received CE (Conformité Européenne) ...
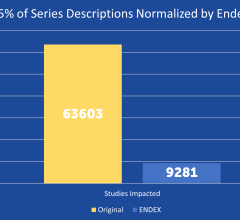
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
November 26, 2017 — Arterys Inc. announced the unveiling of their Arterys MICA platform at the 2017 Radiological Society ...
Samsung Electronics debuted its OmniTom mobile 16-slice computed tomography (CT) scanner at the Radiological Society of North America (RSNA) 2017 Annual Meeting, Nov. 26-Dec. 1 in Chicago. OmniTom received 510(k) U.S. Food and Drug Administration (FDA) clearance for the U.S. market on Aug. 18 of this year.
Lumedx Corp. will present the latest in cardiovascular (CV) imaging, data management and next-generation analytics solutions at the 103rd Scientific Assembly and Annual Meeting of the Radiological Society of North America (RSNA), Nov. 26-Dec. 1 in Chicago. The company said that they will showcase software developed to help heart and vascular hospitals improve outcomes, streamline the delivery of patient care and reduce costs.
November 22, 2017 — Intelerad Medical Systems announced that Diagnostic Centers of America (DCA) has selected Intelerad ...
Carestream Health announced its Carestream Touch Prime Ultrasound Systems have received a 2017 Innovative Technology designation from Vizient Inc., the largest member-driven healthcare performance improvement company in the country. The designation was based on direct feedback from hospital experts who interacted with the Touch Prime Ultrasound Systems at the Vizient Innovative Technology Exchange in Denver.
November 22, 2017 — Paxeramed will showcase its patient-centric imaging exchange, CareRad, at the 2017 Radiological ...

 December 04, 2017
December 04, 2017