Kubtec has received approval to market its XPERT 40 digital specimen radiography device from the FDA. The 510(k) premarket approval notification adds to the CE, CSA, Health Canada and CDRH certifications that have already been obtained for the device.
September 5, 2007 - The FDA has approved Evithrom (human thrombin), a blood-clotting protein used to help control ...
Standard Imaging announced at AAPM the final release of RAy Film Dosimetry Software, a 3D solution for radiation ...
Fujifilm’s APERTO Lucent is a 0.4T mid-field, open MRI system addressing today’s capability and image quality needs ...
NovaPACS 6.6 by NovaRad offers embedded MIP (maximum intensity projection) and MPR (multiplanar reconstruction) tools to ...
September 5, 2007 — RadNet Managed Imaging Services Inc. ("RMIS"), a wholly owned RadNet subsidiary, will provide ...

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
Siemens recently rolled out MVision, an IGRT system that is said to deliver outstanding image quality for accurate ...
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
Fifteen years ago, a CT scan of Nefertiti’s bust stored at Berlin’s Altes Museum, revealed that a second structure was ...
Merge Healthcare recently introduced Merge Ortho, a digital workstation solution that reportedly helps orthopaedic ...
September 5, 2007 — A new technology for cancer detection that eliminates the need for drawing blood has been developed ...
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
September 5, 2007 - Imaging Diagnostic Systems Inc. has installed a CT Laser Mammography (CTLM) system at the Tianjin ...
Toshiba has announced a complete package of post-processing 3D and 4D software solutions for MR, CT, ultrasound and ...
Medrad Inc. introduced at the American Healthcare Radiology Administrators (AHRA) Annual Meeting the XDS, a new extravasation detector technology for the Stellant CT Injection System that, for the first time, directly senses extravasation pooling under the skin during a CT procedure.
Did you know that approximately one-third of all the data in world is created by the healthcare industry and that ...
September 5, 2007 - The FDA has licensed a new vaccine to protect people at high risk of exposure to smallpox during a ...
North American Scientific Inc. announced that its NOMOS Radiation Oncology division will introduce the recently FDA 510 ...
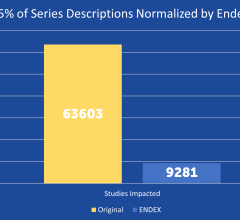
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
Siemens Medical Solutions Ultrasound Division recently introduced the 5.0 release of their ACUSON Antares ultrasound ...
UniWeb PACS, a multimodality Web-based solution from EBM Technologies, has received FDA clearance. EBM UniWeb can ...
September 4, 2007 - Mediware Information Systems Inc. appointed Thomas K. Mann as the company's new chief executive ...
September 4, 2007 - The first few minutes are instrumental in emergency situations and a new portable ultrasound device ...
September 4, 2007 - Agfa HealthCare demonstrated at the European Society of Cardiology Congress 2007 its new IMPAX ...
September 4, 2007 – Philips recently demonstrated its HeartStart MRx monitor/defibrillator that enables paramedics to ...

 September 04, 2007
September 04, 2007