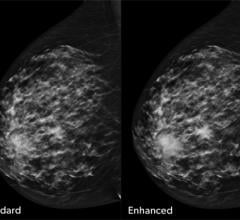
December 19, 2017 — Humanetics Corp. presented data at the annual meetings of the American Society for Radiation ...
December 19, 2017 — IBA (Ion Beam Applications SA) announced that Miami Cancer Institute treated its first patients ...
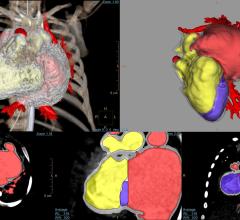
December 19, 2017 — Toshiba Medical, a Canon Group company, introduced the Galan RT Solutions suite of radiation therapy ...
Fujifilm’s APERTO Lucent is a 0.4T mid-field, open MRI system addressing today’s capability and image quality needs ...
iCAD Inc. announced the results of a landmark study that showed the benefits of adjunct intraoperative radiation therapy (IORT) compared to external beam radiation therapy (EBRT) in the treatment of early-stage breast cancer. The analysis demonstrated that IORT could result in direct cost savings for the U.S. healthcare system of more than $630 million over the lifetime of patients diagnosed annually with early-stage breast cancer, as well as significantly benefit patient health by minimizing radiation exposure and offering a better quality of life. The results of the study, which were originally published in the peer-reviewed Cost Effectiveness and Resource Allocation, determined IORT to be the preferred method of treatment.
The U.S. Food and Drug Administration (FDA) announced in a new drug safety communication is this week that it is ...

SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
Over the past couple years covering the news about gadolinium-based contrast agent (GBCA) retention in the brain, skin ...
SPONSORED CONTENT — Fujifilm’s latest CT technology brings exceptional image quality to a compact and user- and patient ...
A post-game roundup by ITN Contributing Editor Greg Freiherr and ITN Editor Dave Fornell on the trends and new tech seen ...
At the 2017 Radiological Society of North America (RSNA) Annual Meeting, GE Healthcare and NVIDIA announced a series of imaging equipment advances powered by NVIDIA’s artificial intelligence (AI) computing platform. The announcements included the new Revolution Frontier computed tomography (CT) system, advancements to the Vivid E95 4-D Ultrasound and development of GE Healthcare’s Applied Intelligence analytics platform.
Optellum, a high-tech startup in lung cancer and machine learning, and Mirada Medical recently announced a partnership to accelerate the deployment of deep learning technology to market. This collaboration combines the deep technology and clinical expertise of Optellum with the software platforms and rapid development know-how of Mirada. They have partnered to address what they call a huge and growing problem in lung cancer diagnosis: the management of patients with indeterminate pulmonary nodules.
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
December 14, 2017 — iCAD Inc. recently announced that customer adoption of its PowerLook Tomo Detection is gaining ...
Philips showcased the company’s latest magnetic resonance (MR) imaging solutions at the Radiological Society of North America’s 2017 annual meeting, Nov. 26-Dec. 1 in Chicago. This includes their newest MR system, MR Prodiva 1.5T (which is still U.S. Food and Drug Administration [FDA] 510(k) pending) that provides enhanced clinical performance, workflow and patient experience. Philips also unveiled new solutions to drastically reduce exam times and elevate neuro-oncology.
Siemens Healthineers introduced four new computed tomography (CT) scanners across two platforms at the 2017 Radiological Society of North America (RSNA) Annual Meeting, Nov. 26-Dec. 1 in Chicago. The two new scanners in the Somatom go. Platform — Somatom go.All and Somatom go.Top — expand the mobile workflow into advanced fields such as cardiology and CT-guided intervention. With its new high-end systems for single- and dual-source imaging — Somatom Edge Plus and Somatom Force — Siemens Healthineers is introducing the new FAST (Fully Assisting Scanner Technologies) Integrated Workflow with the new FAST 3-D Camera.
Did you know that approximately one-third of all the data in world is created by the healthcare industry and that ...
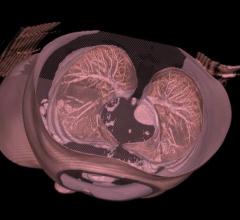
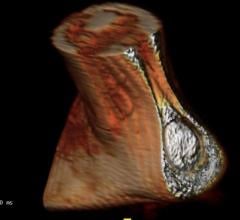
Toshiba Medical, a Canon Group company, introduced its new Aquilion Prime SP computed tomography (CT) system at the 2017 Radiological Society of North America (RSNA) Annual Meeting, Nov. 26-Dec. 1 in Chicago. The CT system features advanced imaging technology from Toshiba Medical’s high-end CT systems, giving clinicians the right balance between image quality and lower dose for every patient from pediatric to bariatric.
Philips announced the debut of IntelliSpace Portal 10, the latest edition of its comprehensive, advanced visualization and quantification platform, at the 2017 Radiological Society of North America (RSNA) Annual Meeting, Nov. 26-Dec. 1 in Chicago. This next generation features enhancements across the solution, with a particular focus on oncology to provide radiologists with an improved full set of applications and workflows to support the reading and follow-up of complex oncology cases. IntelliSpace Portal 10 also includes a new 3-D modeling application and has been expanded with the DynaCAD Prostate and Breast solutions through integration with InVivo.
SPONSORED CONTENT — EnsightTM 2.0 is the newest version of Enlitic’s data standardization software framework. Ensight is ...
Emanuel Kanal, M.D., director of MRI services and professor of radiology and neuroradiology at the University of ...
In the run-up to RSNA 1999, General Electric unveiled its 0.7T OpenSpeed. It was easy to be impressed. The conference ...
AI Visualize Inc. debuted its new cloud-based artificial intelligence (A.I.) -based image analysis and viewing platform at the 2017 Radiological Society of North America (RSNA) Annual Meeting, Nov. 26-Dec. 1 in Chicago. The technology relies on evolution and deep learning algorithms to assess imaging data for diagnostically valuable information not readily apparent through conventional analysis, and transmits detailed 3-D/2-D renderings to users on any internet-enabled device. The technology assists physicians in making the most of today’s data-intensive images for more accurate and precise diagnoses. The advanced interactive platform supports X-ray, ultrasound, computed tomography (CT), magnetic resonance imaging (MRI), 3-D tomosynthesis and digital pathology images, and provides a full range of relevant virtual image analysis tools.
GE Healthcare announced U.S. FDA 510(k) clearance of its new Air Technology, what it calls an industry-first suite of radiofrequency (RF) coils that enables total freedom in coil positioning and handling during a magnetic resonance imaging (MRI) scan. The new coil design is 60 percent lighter than conventional coils, benefiting both patients and technologists, offers greater flexibility in all axes to help conform to patients’ anatomies and fits all patient ages, sizes and shapes.
J. Anthony Seibert, Ph.D., professor and chair of informatics, University of California Davis, describes the security ...
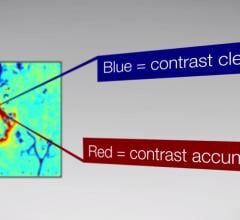
Brainlab announced U.S. Food and Drug Administration (FDA) clearance of its Contrast Clearance Analysis methodology, developed at Sheba Medical Center in Tel-Hashomer, Israel, with technology provided by Brainlab. The software analyzes magnetic resonance (MR) images to differentiate regions of efficient contrast clearance from regions with contrast accumulation in most cranial tumor patients to provide additional insight into post-treatment tumor characteristics.


 December 19, 2017
December 19, 2017